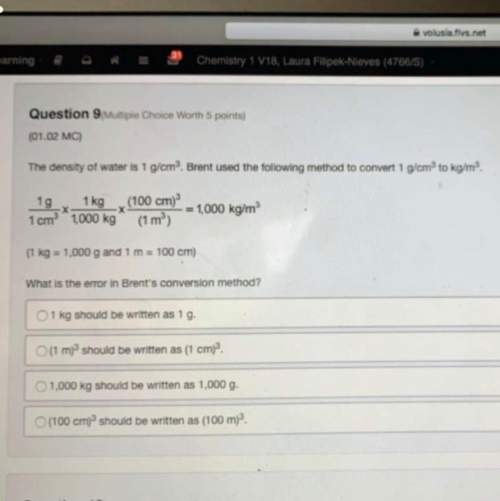
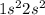Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 18:30
How many moles of lead are in 1.50 x 10^12 atoms of lead? could you explain the answer as well and not just give it to me i am refreshing for finals and i need to know how to do it
Answers: 3


Chemistry, 22.06.2019 21:30
If you burn 46.6 g of hydrogen and produce 416 g of water, how much oxygen reacted
Answers: 3
You know the right answer?
Beryllium (be): 1sc2sd, what's c and d?...
Questions



Chemistry, 27.01.2021 22:50




Mathematics, 27.01.2021 22:50

Mathematics, 27.01.2021 22:50






Mathematics, 27.01.2021 22:50

Mathematics, 27.01.2021 22:50

Mathematics, 27.01.2021 22:50




Chemistry, 27.01.2021 22:50