Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 17:10
A+b→2c when the reaction begins, the researcher records that the rate of reaction is such that 1 mole of a is consumed per minute. after making changes to the reaction, the researcher notes that 2 moles of a are consumed per minute. what change could the researcher have made to effect this change?
Answers: 1


Chemistry, 22.06.2019 10:30
Acompound has a molar mass of 92.02 grams/mole, and its percent composition is 30.4% nitrogen (n) and 69.6% oxygen (o). what is its molecular formula? a. n2o4 b. no2 c. n2o d. n4o2
Answers: 1

Chemistry, 22.06.2019 12:30
If 22.5 liters of oxygen reacted with excess of hydrogen, how many liters of water vapor could be produced?
Answers: 3
You know the right answer?
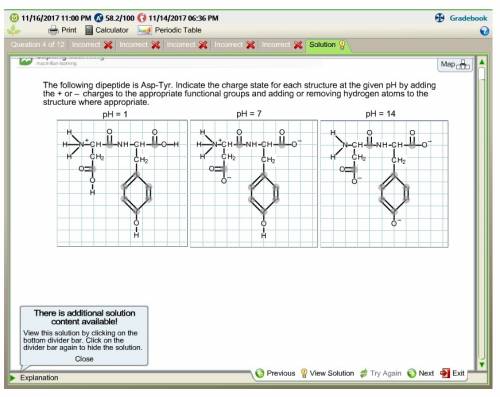
The following dipeptide is asp-tyr. indicate the charge state for each structure at the given ph by...
Questions

Mathematics, 05.09.2020 01:01


Chemistry, 05.09.2020 01:01

Social Studies, 05.09.2020 01:01




Mathematics, 05.09.2020 01:01


Mathematics, 05.09.2020 01:01


Social Studies, 05.09.2020 01:01




Mathematics, 05.09.2020 01:01