
Chemistry, 16.07.2019 11:50 gameranonymous266
Describe a method you could use to obtain crystals of potassium nitrate from potassium nitrate solution. you should include an explanation of how the crystals are produced from the solution.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 10:40
Ammonia and oxygen react to form nitrogen monoxide and water, like this: 4nh3 (g) + 5o2 (g) → 4no (g) + 6h2o (g) also, a chemist finds that at a certain temperature the equilibrium mixture of ammonia, oxygen, nitrogen monoxide, and water has the following composition: compound pressure at equilibrium nh3 65.1atm o2 31.3atm no 62.7atm h2o 65.8atm compound pressure at equilibrium nh3 65.3 atm o2 7.79 atm no 12.1 atm h2o 65.8 atm calculate the value of the equilibrium constant kp for this reaction. round your answer to 2 significant
Answers: 2

Chemistry, 22.06.2019 10:50
How many liters of oxygen gas, at standard temperature and pressure, will react with 35.8 grams of iron metal? 4 fe (s) + 3 o₂ (g) → 2 fe₂o₃ (s)
Answers: 2

Chemistry, 22.06.2019 16:30
At 20°c, a sample of h2o liquid and a sample of co2 gas each have the same average kinetic energy. why is one a liquid and the other a gas at this temperature?
Answers: 1

Chemistry, 22.06.2019 17:00
Reduction is a reaction which results in a in electrons and a in positive charge of the atom or ion 1) a- loss 1) b- gain 2) a-increase 2) b-decrease
Answers: 1
You know the right answer?
Describe a method you could use to obtain crystals of potassium nitrate from potassium nitrate solut...
Questions



History, 23.08.2019 16:30

History, 23.08.2019 16:30





Geography, 23.08.2019 16:30


Mathematics, 23.08.2019 16:30




English, 23.08.2019 16:30

Mathematics, 23.08.2019 16:30



Chemistry, 23.08.2019 16:30

Mathematics, 23.08.2019 16:30

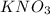 . The shape of potassium nitrate is orthorhombic or regular prism.
. The shape of potassium nitrate is orthorhombic or regular prism.

