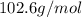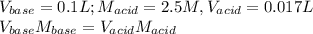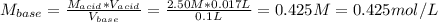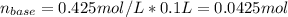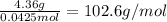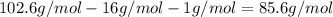Chemistry, 13.07.2019 10:20 Kennethabrown09
A4.36 g sample of an unknown alkali metal hydroxide is dissolved in 100.0 ml of water. an acid-base indicator is added and the resulting solution is titrated with 2.50 m hcl (aq) solution. the indicator changes color signaling that the equivalent point has been reached after 17.0 ml of the hydrochloric acid solution has been added. (a) what is the molar mass of the metal hydroxide? (b) what is the identity of the metal cation?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 23:00
What is the volume of the fluid in the graduated cylinder with accuracy and measured to the correct degree of precision? 41.2 ml 42.0 ml 41.23 ml 41.89 ml
Answers: 1

Chemistry, 22.06.2019 07:30
Aradio signal from a gps satellite take only about 0.067 seconds to reach a gps reciever. if the speed of light is about 300,000km/s, then approximately how far away is the reciever from from the satellite?
Answers: 1

Chemistry, 22.06.2019 13:00
Is 9 correct? and can someone me with 10? it’s due tomorrow, you
Answers: 1

Chemistry, 22.06.2019 22:30
Consider a culture medium on which only gram-positive organisms such as staphylococcus aureus colonies can grow due to an elevated nacl level. a yellow halo surrounds the growth, indicating the bacterium fermented a sugar in the medium, decreasing the ph as a result and changing the color of a ph indicator chemical. this type of medium would be referred to as a differential and enrichment culture.
Answers: 2
You know the right answer?
A4.36 g sample of an unknown alkali metal hydroxide is dissolved in 100.0 ml of water. an acid-base...
Questions

Mathematics, 21.10.2019 20:30


Biology, 21.10.2019 20:30

Chemistry, 21.10.2019 20:30



Mathematics, 21.10.2019 20:30


Chemistry, 21.10.2019 20:30




History, 21.10.2019 20:30


History, 21.10.2019 20:30



Biology, 21.10.2019 20:30

Biology, 21.10.2019 20:30