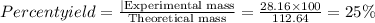Chemistry, 12.07.2019 21:40 jsharma57p7enrw
Give the percent yield when 28.16 g of co2 are formed from the reaction of 8.000 moles of c8h18 with 4.000 moles of o2. 2 c8h18 + 25 o2 → 16 co2 + 18 h2o

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 07:00
The boiling point of propanoic acid is higher than that of 1-butanol because: propanoic acid has a higher molecular weight than 1-butanol. propanoic acid is more soluble in water than 1-butanol. propanoic acid is a better hydrogen bond donor than 1-butanol. propanoic acid forms hydrogen bonded dimers and 1-butanol does not. 1-butanol forms hydrogen bonded dimers and propanoic acid does not.
Answers: 2

Chemistry, 22.06.2019 13:30
Apush or pull that moves or changes and object when to objects touch
Answers: 2

Chemistry, 22.06.2019 17:20
The small bags of silica gel you often see in a new shoe box are placed there to control humidity. despite its name, silica gel is a solid. it is a chemically inert, highly porous, amorphous form of sio2. because water vapor readily adsorbs onto the surface of silica gel, it acts as a desiccant. despite not knowing mechanistic details of the adsorption of water onto silica gel, from the information provided you should be able to make an educated guess about the thermodynamic characteristics of the process. predict the signs for δg, δh, and δs for the adsorption of water.
Answers: 2

Chemistry, 22.06.2019 18:00
How does climate change cause the ocean's thermohaline current to slow down?
Answers: 3
You know the right answer?
Give the percent yield when 28.16 g of co2 are formed from the reaction of 8.000 moles of c8h18 with...
Questions



Social Studies, 12.10.2019 12:30

English, 12.10.2019 12:30


History, 12.10.2019 12:30

History, 12.10.2019 12:30

Social Studies, 12.10.2019 12:30

Physics, 12.10.2019 12:30


History, 12.10.2019 12:30

Health, 12.10.2019 12:30

Chemistry, 12.10.2019 12:30



Mathematics, 12.10.2019 12:30

English, 12.10.2019 12:30

Mathematics, 12.10.2019 12:30

Mathematics, 12.10.2019 12:30

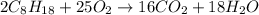
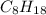 , then 4 moles of oxygen will react with
, then 4 moles of oxygen will react with 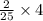 moles of
moles of 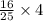 moles of carbon-dioxide gas that is 2.56 moles.
moles of carbon-dioxide gas that is 2.56 moles.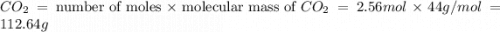
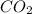 = 28.16 g
= 28.16 g