
Chemistry, 10.07.2019 10:40 taysomoneyyy
Combustion of 25.0 g of a hydrocarbon produces 86.5 g of co2. what is the empirical formula of the compound?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 10:30
Balance and in which category does it fit in? single or double displacement or synthesis or decomposition? (a) k2 o → k + o2 (b) na + i2 → nai (c) cu(no3 )2 + naoh → cu(oh)2 + nano3 (d) kclo3 → kcl + o2 (e) ca(no3 )2 + hbr → cabr2 + hno3 (f) sn(oh)2 → sno + h2 o (g) p4 + n2 o → p4 o6 + n2 (h) fe + al2 (so4 )3 → feso4 + al (i) alcl3 + na2 co3 → al2 (co3 )3 + nacl (j) c3 h6 + o2 → co2 + h2 o
Answers: 1


Chemistry, 22.06.2019 12:00
Ican determine the molar mass of an element by looking on the under the atomic mass for the element. for example the molar mass of phosphorus is 30.974 grams/mole. avogadro’s number tells me the amount of representative particles in 1 mole of any substance. this means 12.011 gram sample of carbon and a 32.0 gram sample of sulfur have the same number of atoms.
Answers: 1

You know the right answer?
Combustion of 25.0 g of a hydrocarbon produces 86.5 g of co2. what is the empirical formula of the c...
Questions




Mathematics, 21.11.2020 01:10

Mathematics, 21.11.2020 01:10



English, 21.11.2020 01:10

Mathematics, 21.11.2020 01:10



Health, 21.11.2020 01:10

Mathematics, 21.11.2020 01:10

English, 21.11.2020 01:10

Mathematics, 21.11.2020 01:10

History, 21.11.2020 01:10

Mathematics, 21.11.2020 01:10

Mathematics, 21.11.2020 01:10

Mathematics, 21.11.2020 01:10

Mathematics, 21.11.2020 01:10

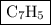 .
.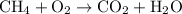
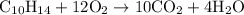
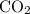 is formed as a product during combustion reactions.
is formed as a product during combustion reactions.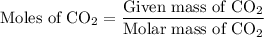 ...... (1)
...... (1)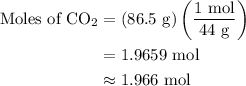
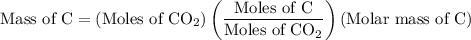 ...... (2)
...... (2)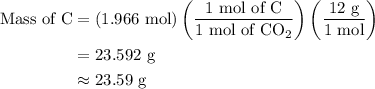
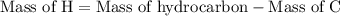 ...... (3)
...... (3)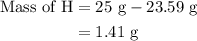
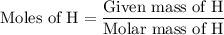 ...... (4)
...... (4)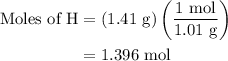
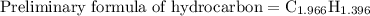

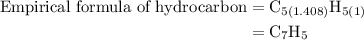
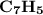 .
.


