Chemistry, 10.07.2019 02:50 lightskinbaby2
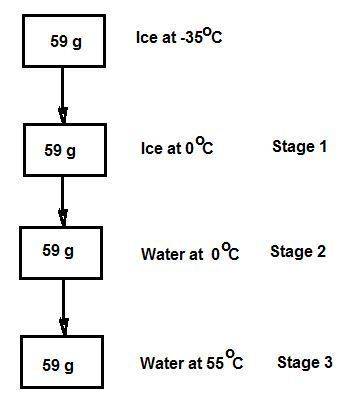
Consider the specific heats h2o(s) = 2.09 j/g · ◦c, h2o (ℓ) = 4.18 j/g · ◦c, and h2o(g) = 2.03 j/g · ◦c. the heat of fusion for water is 334 j/g and its heat of vaporization is 2260 j/g. calculate the amount of heat required to convert 59 g of ice at −35◦c completely to liquid water at 55◦c. answer in units of kj.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 17:30
Observation and experimentation have led many scientists to accept a theory about the origin of the universe. this theory is called the big bang theory. scientific evidence collected and observed by scientists around the world suggests that the universe is ever expanding from a hot and dense initial state. what makes this a scientific theory? (2 points)
Answers: 2


Chemistry, 23.06.2019 04:00
What changes occur in the reaction indicated by the equation? check all that apply. the hydrogen nucleus loses protons. the oxygen nucleus gains protons. the bond in h2 is broken, and new bonds are formed between hydrogen and oxygen atoms. each electron associated with a hydrogen atom is shared with an oxygen atom.
Answers: 3

Chemistry, 23.06.2019 05:00
If 15 drops of ethanol from a medicine dropper weigh 0.60g, how many drops does it takes from a dropper to dispense 1.0ml of ethanol? density of ethanol is ethanol is 0.80g/ml.
Answers: 2
You know the right answer?
Consider the specific heats h2o(s) = 2.09 j/g · ◦c, h2o (ℓ) = 4.18 j/g · ◦c, and h2o(g) = 2.03 j/g ·...
Questions


Chemistry, 22.03.2021 18:00

Social Studies, 22.03.2021 18:00

Mathematics, 22.03.2021 18:00


Geography, 22.03.2021 18:00

Computers and Technology, 22.03.2021 18:00

Biology, 22.03.2021 18:00

Mathematics, 22.03.2021 18:00


Mathematics, 22.03.2021 18:00

English, 22.03.2021 18:00

Mathematics, 22.03.2021 18:00


Arts, 22.03.2021 18:00

Mathematics, 22.03.2021 18:00

Mathematics, 22.03.2021 18:00

Mathematics, 22.03.2021 18:00

Mathematics, 22.03.2021 18:00

Mathematics, 22.03.2021 18:00