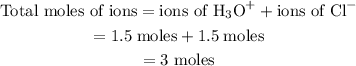If you have an aqueous solution that contains 1.5 moles of hcl, how many moles of ions are in the solution? (a) 1.0, (b) 1.5, (c) 2.0, (d) 2.5, (e) 3.if you have an aqueous solution that contains 1.5 moles of hcl, how many moles of ions are in the solution? (a) 1.0, (b) 1.5, (c) 2.0, (d) 2.5, (e) 3.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 20:30
We are hoping to create 5.72 grams of glucose. the plant was given 4.75 liters of co2 and 2.81 g of h20. which reactant was the limiting reagent? how much excess mass did we have of the other reactant?
Answers: 2

Chemistry, 22.06.2019 20:30
Select all the correct answers.which compounds have the empirical formula ch20? (multiple answers)a.c2h4o2b.c3h603c.ch2o2d.c5h1005e.c6h1206
Answers: 2


You know the right answer?
If you have an aqueous solution that contains 1.5 moles of hcl, how many moles of ions are in the so...
Questions


English, 05.11.2020 19:20



English, 05.11.2020 19:20

English, 05.11.2020 19:20










History, 05.11.2020 19:20

SAT, 05.11.2020 19:20


Computers and Technology, 05.11.2020 19:20

Mathematics, 05.11.2020 19:20

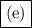 . The number of moles of ion in the solution is
. The number of moles of ion in the solution is 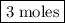
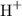 ions or can accept electrons from the electron-rich species. The acids can be classified into two types as follows:
ions or can accept electrons from the electron-rich species. The acids can be classified into two types as follows: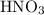 and
and 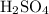 . The general dissociation reaction of a strong acid is as follows:
. The general dissociation reaction of a strong acid is as follows: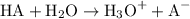
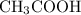 , HCOOH and HCN. The general dissociation reaction of a weak acid is as follows:
, HCOOH and HCN. The general dissociation reaction of a weak acid is as follows: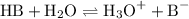
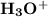 and
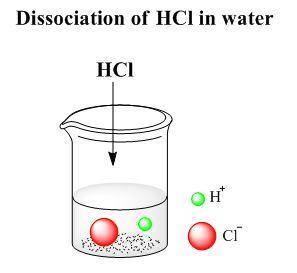
and 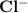 ions. The dissociation reaction of HCl is as follows:
ions. The dissociation reaction of HCl is as follows: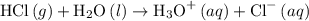
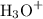 ion and one
ion and one 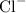 mole of ion. Therefore 1.5 mol of HCl will dissociate to give 1.5 mol of
mole of ion. Therefore 1.5 mol of HCl will dissociate to give 1.5 mol of