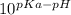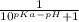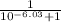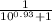Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 19:30
Estimate the molar mass of the gas that effuses at 1.6 times the effusion rate of carbon dioxide.
Answers: 1

Chemistry, 22.06.2019 22:30
You just calculated that the heat of fusion for chloromethane is 6400 j/mol. the heat of fusion for hydrogen is 120 j/mol.? which of the following account for this difference? more than one correcta. chloromethane can absorb more energy at the same temperature. b. hydrogen has stronger intermolecular forces than chloromethane. c. hydrogen molecules can pack more closely than chloromethane molecules. d. chloromethane experiences dipole-dipole interactions. e. chloromethane has a higher molar mass than hydrogen.
Answers: 3

You know the right answer?
The pka of the α-carboxyl group of glutamine is 2.17, and the pka of its α-amino group is 9.13. calc...
Questions

Mathematics, 15.01.2021 03:40





Mathematics, 15.01.2021 03:40

Mathematics, 15.01.2021 03:40




History, 15.01.2021 03:40

Mathematics, 15.01.2021 03:40

English, 15.01.2021 03:40

Mathematics, 15.01.2021 03:40


Mathematics, 15.01.2021 03:40

Mathematics, 15.01.2021 03:40


Mathematics, 15.01.2021 03:40

Mathematics, 15.01.2021 03:40