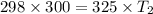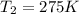Chemistry, 05.07.2019 09:00 natalie2sheffield
The pressure in a car tire is 298 kilopascals at 300 kelvin. after a long drive, the pressure becomes 325 kilopascals. which law will you use to calculate the new temperature in the tire assuming that the volume is constant?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 23:20
Harvey kept a balloon with a volume of 348 milliliters at 25.0˚c inside a freezer for a night. when he took it out, its new volume was 322 milliliters, but its pressure was the same. if the final temperature of the balloon is the same as the freezer’s, what is the temperature of the freezer? the temperature of the freezer is kelvins.
Answers: 2

Chemistry, 23.06.2019 00:10
In as 1°, 2°, 3°, or 4°. be to . : °b: °c: °d: ° : °b: °c: °d: ° : °b: °c: °d: °e: °f: °g: °h: ° : °b: °c: °d: °e: °f: °g: °h: °i: °
Answers: 3

Chemistry, 23.06.2019 00:30
When did stem cell research become known ? who discovered stem cell? what experiments or studies have been conducted so far?
Answers: 3

Chemistry, 23.06.2019 05:30
For the reaction i2(g)+br2(g)←−→2ibr(g), kc=280 at 150 ∘c. suppose that 0.450 mol ibr in a 2.00-l flask is allowed to reach equilibrium at 150 ∘c. what is the equilibrium concentration of 2ibr, i2, br2
Answers: 1
You know the right answer?
The pressure in a car tire is 298 kilopascals at 300 kelvin. after a long drive, the pressure become...
Questions


Mathematics, 12.11.2020 07:30


Biology, 12.11.2020 07:30




Mathematics, 12.11.2020 07:30

Biology, 12.11.2020 07:30

History, 12.11.2020 07:30


Arts, 12.11.2020 07:30

Spanish, 12.11.2020 07:30


Biology, 12.11.2020 07:30

Mathematics, 12.11.2020 07:30

Mathematics, 12.11.2020 07:30

Mathematics, 12.11.2020 07:30

Chemistry, 12.11.2020 07:30

English, 12.11.2020 07:30

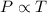 (At constant volume and number of moles)
(At constant volume and number of moles)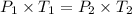
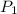 = initial pressure of gas = 298 kPa
= initial pressure of gas = 298 kPa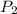 = final pressure of gas = 325 kPa
= final pressure of gas = 325 kPa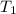 = initial temperature of gas = 300 K
= initial temperature of gas = 300 K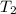 = final temperature of gas = ?
= final temperature of gas = ?