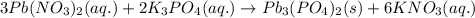Chemistry, 04.07.2019 22:50 natalie407888
When aqueous solutions of lead(ii) nitrate (pb(no3)2) and potassium phosphate (k3po4) are mixed, the products are solid lead(ii) phosphate and aqueous potassium nitrate. write the balanced chemical equation for this reaction. (include states-of-matter under the given conditions in your answer. use the lowest possible whole number

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 01:00
Agas occupies 475 cm^3 at 313k. find its volume at 367k. you must show all of your work to receive credit. be sure to identify which of the gas laws you will be using
Answers: 2

Chemistry, 22.06.2019 07:20
Which of these conditions most likely produces an unstable isotope?
Answers: 1

Chemistry, 22.06.2019 12:00
What is the lowest number energy level where a d sublevel is found
Answers: 1

Chemistry, 23.06.2019 02:00
When an experimenter draws a conclusion that he assumes will apply to all situations set up similarly to his test situation, even though he cannot possibly have examined all possible test scenarios, the experimenter is using deductive reasoning inductive reasoning abductive reasoning subjective reasoning
Answers: 1
You know the right answer?
When aqueous solutions of lead(ii) nitrate (pb(no3)2) and potassium phosphate (k3po4) are mixed, the...
Questions




Mathematics, 17.12.2019 18:31



Physics, 17.12.2019 18:31


Chemistry, 17.12.2019 18:31

Biology, 17.12.2019 18:31

Mathematics, 17.12.2019 18:31




Mathematics, 17.12.2019 18:31

Health, 17.12.2019 18:31