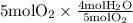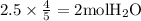Chemistry, 04.07.2019 21:50 Jasmine3864
The combustion of propane (c3h8) produces co2 and h2o: c3h8 (g) + 5o2 (g) → 3co2 (g) + 4h2o (g) the reaction of 2.5 mol of o2 with 4.6 mol of c3h8 will produce mol of h2o.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 11:00
What is the temperature of 0.750 mol of a gas stored in a 6,850 ml cylinder at 2.21 atm? . 2.95 k 5.24 k 138 k 246 k
Answers: 3


Chemistry, 23.06.2019 01:00
Na chemical reaction, activation energy increases the of the reactants. this outcome causes the particles to collide, which results in the of new products.
Answers: 2

Chemistry, 23.06.2019 03:00
Achemical equilibrium between gaseous reactants and products is shown. n2(g) + 3h2(g) ⇌ 2nh3(g) how will the reaction be affected if the pressure on the system is increased? it will shift toward the reactant side as there is lower pressure on the reactant side. it will shift toward the product side as there is higher pressure on the product side. it will shift toward the reactant side as there are a greater number of moles of gas on the reactant side. it will shift toward the product side as there are a fewer number of moles of gas on the product side.
Answers: 2
You know the right answer?
The combustion of propane (c3h8) produces co2 and h2o: c3h8 (g) + 5o2 (g) → 3co2 (g) + 4h2o (g) the...
Questions

English, 23.02.2021 22:20


History, 23.02.2021 22:20

English, 23.02.2021 22:20

Mathematics, 23.02.2021 22:20

Mathematics, 23.02.2021 22:20



Mathematics, 23.02.2021 22:20

Geography, 23.02.2021 22:20

Mathematics, 23.02.2021 22:20

Spanish, 23.02.2021 22:20






Mathematics, 23.02.2021 22:20


Mathematics, 23.02.2021 22:20

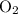 with 4.6 mol
with 4.6 mol 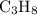 will produce 2mol
will produce 2mol 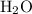 .
.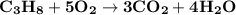
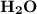 produced will be,
produced will be,