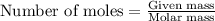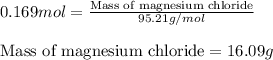Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 03:30
At a temperature of 393 k, the temperature of a sample of nitrogen is 1.07 atm what will the pressure be at a temperature of 478 k
Answers: 1

Chemistry, 22.06.2019 22:00
Plz ill give u brainliest which of the following steps is not likely to take place during cellular respiration? a.oxygen combines with carbon of simple sugar. b. energy molecule transfers energy to cells. d. energy is used up.
Answers: 3

Chemistry, 22.06.2019 22:30
Molecular iodine, i2(g), dissociates into iodine atoms at 625 k with a first-order rate constant of 0.271 s−1. part a part complete what is the half-life for this reaction?
Answers: 3

Chemistry, 23.06.2019 06:00
Nthis lab, you will do experiments to identify types of changes. using the question format you learned (shown above), write an investigative question that you can answer by doing these experiments
Answers: 3
You know the right answer?
Consider the reaction of magnesium metal with hydrochloric acid to produce magnesium chloride and hy...
Questions




Mathematics, 20.07.2019 00:00

History, 20.07.2019 00:00

Biology, 20.07.2019 00:00



Social Studies, 20.07.2019 00:00





Social Studies, 20.07.2019 00:00

Computers and Technology, 20.07.2019 00:00


Mathematics, 20.07.2019 00:00



Mathematics, 20.07.2019 00:00


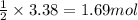 of magnesium.
of magnesium.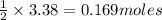 of magnesium chloride.
of magnesium chloride.