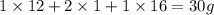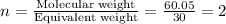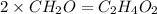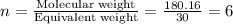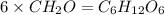The empirical formula for two compounds that have very different properties (one tastes very sour and the other very sweet) is ch2o. if the molar mass of compound a is 60.05 g/mol and compound b is 180.16 g/mol, what are the molecular formulas for these compounds, respectively?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:00
During chemistry class, carl performed several lab tests on two white solids. the results of three tests are seen in the data table. based on this data, carl has concluded that substance b must have bonds.
Answers: 2



Chemistry, 22.06.2019 23:40
The kw for water at 0 °c is 0.12× 10–14 m2. calculate the ph of a neutral aqueous solution at 0 °c.
Answers: 2
You know the right answer?
The empirical formula for two compounds that have very different properties (one tastes very sour an...
Questions





Mathematics, 17.04.2020 20:04

History, 17.04.2020 20:04









Mathematics, 17.04.2020 20:04

History, 17.04.2020 20:04


Social Studies, 17.04.2020 20:04

Mathematics, 17.04.2020 20:04

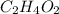
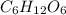
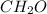 , empirical weight is =
, empirical weight is =