The type of bond between atoms in a molecule of co2 (electronegativity difference = ~1)
a. hyd...

Chemistry, 19.08.2019 18:30 quintinjerome
The type of bond between atoms in a molecule of co2 (electronegativity difference = ~1)
a. hydrogen bond
b. ionic bond
c. polar covalent bond
d. pure covalent bond
e. metallic bond

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 13:00
Read the given expression. x = number of protons − number of core electrons which of the following explains the identity of x and its trends across a period? x is the effective nuclear charge, and it remains constant across a period. x is the screening constant, and it remains constant across a period. x is the effective nuclear charge, and it increases across a period. x is the screening constant, and it increases across a period.
Answers: 1

Chemistry, 22.06.2019 03:30
Each pair of clay balls represents to planetesimals if each plane test molluscum pound of the same material and is separated by the same distance which pair experiences the greatest gravitational attraction
Answers: 2

Chemistry, 22.06.2019 12:20
Which is an example of the practical pursuit of alchemy? a. forming perfect substances. b. transforming base metals. c. developing metalworking techniques. d. linking spiritual characteristics with material substances.
Answers: 1

Chemistry, 22.06.2019 14:20
Which statement explains why the bonds between non metals tend to be covalent? the bonds are found to be nondirectional they have large differences in electronegativity they have small differences in electronegativity they have ions that produce an electrostatic pull
Answers: 1
You know the right answer?
Questions


Mathematics, 26.10.2019 18:43








Geography, 26.10.2019 19:43

Mathematics, 26.10.2019 19:43


English, 26.10.2019 19:43


History, 26.10.2019 19:43

Mathematics, 26.10.2019 19:43




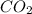 , is a pure covalent bond.
, is a pure covalent bond. 

