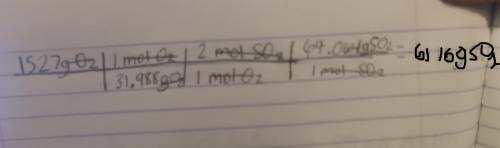Chemistry, 11.03.2022 21:20 singletaryshawp31cmx
2 SO2(g) + O2(g) + 2 H2O(ℓ) −→ 2 H2SO4(ℓ)
What mass in grams of SO2 is needed to
react with 1527 g of O2?

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 18:30
Amass of 100.0 g of solute is dissolved in water so that 850. ml of a 0.7500 m solution has been prepared. what is the molar mass of the solute?
Answers: 2

Chemistry, 22.06.2019 07:10
An experimental procedure requires a 10 ml of acid to be dissolved
Answers: 2

Chemistry, 22.06.2019 09:10
Select the correct answer from each drop-down menu.describe what happens to a carbon-11 atom when it undergoes positron emission.the decay of a carbon-11 atom _1_, and this causes it to emit _2_.options for 1: > changes a neutron into a proton> changes a proton into a neutron> is hit with a neutron> reconfigures its protons and neutronsoptions for 2: > a negatively charged electron-sized particle> a positively charged election-sized particle> two atoms and several neutrons> two neutrons and two protons
Answers: 3

Chemistry, 22.06.2019 14:20
You have a liquid that exhibits diltancy. you want to pour it from a bottle. what should you do to the bottle before pouring
Answers: 1
You know the right answer?
2 SO2(g) + O2(g) + 2 H2O(ℓ) −→ 2 H2SO4(ℓ)
What mass in grams of SO2 is needed to
react with...
react with...
Questions


Mathematics, 28.09.2021 14:00



Physics, 28.09.2021 14:00


Mathematics, 28.09.2021 14:00

Mathematics, 28.09.2021 14:00


Biology, 28.09.2021 14:00

Mathematics, 28.09.2021 14:00

Mathematics, 28.09.2021 14:00


Medicine, 28.09.2021 14:00

Chemistry, 28.09.2021 14:00

Chemistry, 28.09.2021 14:00

English, 28.09.2021 14:00

English, 28.09.2021 14:00


Mathematics, 28.09.2021 14:00