
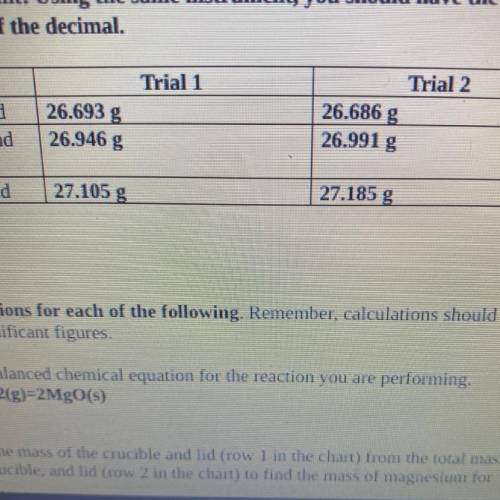
4. Magnesium is the limiting reactant in this experiment. Calculate the theoretical
yield of Mgo for each trial.
. Trial 1:
• Trial 2:
5. Determine the percent yield of Mgo for your experiment for each trial.
Trial 1:
• Trial 2:
6. Determine the average percent yield of Mgo for the two trials.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 03:10
Agas diffuses 1/7 times faster than hydrogen gas (h2). what is the molar mass of the gas? 100.10 g/mol 98.78 g/mol 86.68 g/mol 79.98 g/mol
Answers: 3


Chemistry, 23.06.2019 02:00
When an experimenter draws a conclusion that he assumes will apply to all situations set up similarly to his test situation, even though he cannot possibly have examined all possible test scenarios, the experimenter is using deductive reasoning inductive reasoning abductive reasoning subjective reasoning
Answers: 1

Chemistry, 23.06.2019 03:00
Determine type of reaction & predict the product c3h12+o2 =
Answers: 1
You know the right answer?
4. Magnesium is the limiting reactant in this experiment. Calculate the theoretical
yield of Mgo f...
Questions


History, 14.10.2019 15:10




Social Studies, 14.10.2019 15:10

Mathematics, 14.10.2019 15:10

Mathematics, 14.10.2019 15:10


Mathematics, 14.10.2019 15:10



Physics, 14.10.2019 15:10




History, 14.10.2019 15:10


Computers and Technology, 14.10.2019 15:10

Mathematics, 14.10.2019 15:10



