
Chemistry, 16.02.2022 06:50 SallyMarquez1201
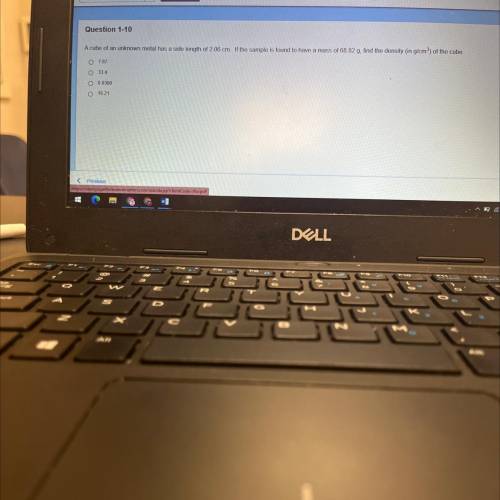
A cube of an unknown metal has a side length of 2.06 cm. If the sample is found to have a mass of 68.82 g, find the density (in g/cm²) of the cube
O 7.87
0 33.4
0 0.0300
16.21

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 18:00
Sylvanite is a mineral that contains 28.0% gold by mass. how much sylvanite would you need to dig up to obtain 77.0 g of gold? explain how you got your answer and the steps you took. you
Answers: 3

Chemistry, 22.06.2019 11:40
Effect of rotenone and antimycin a on electron transfer rotenone, a toxic natural product from plants, strongly inhibits nadh dehydrogenase of insect and fish mitochondria. antimycin a, a toxic antibiotic, strongly inhibits the oxidation of ubiquinol. (a) explain why rotenone ingestion is lethal to some insect and fish species. (b) explain why antimycin a is a poison. (c) given that rotenone and antimycin a are equally effective in blocking their respective sites in the electron-transfer chain, which would be a more potent poison? explain.
Answers: 3

Chemistry, 23.06.2019 00:00
How is the way a mixture is combined different from how a compound is combined?
Answers: 3

Chemistry, 23.06.2019 01:00
Iron (fe) reacts with copper sulfate (cuso4) to form iron (ii) sulfate. in this reaction, cu2+ gains electrons to form cu. which statement is true about this reaction? fe(s) + cuso4(aq) → feso4(aq) + cu(s)
Answers: 3
You know the right answer?
A cube of an unknown metal has a side length of 2.06 cm. If the sample is found to have a mass of 68...
Questions


Computers and Technology, 20.08.2019 23:20




Computers and Technology, 20.08.2019 23:20






Computers and Technology, 20.08.2019 23:20




Computers and Technology, 20.08.2019 23:20



English, 20.08.2019 23:20

History, 20.08.2019 23:20



