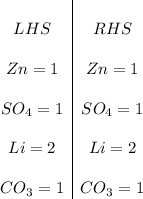Balance the equation:THANK U IF U FIND IT!!
...

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 18:00
When the following equation is balanced using the smallest possible integers, what is the coefficent of oxygen gas? c7h16o(g) + o2(g) → co2(g) + h2o(g) -1 -5 -8 -16 -21
Answers: 3

Chemistry, 22.06.2019 06:30
The following reaction shows sodium carbonate reacting with calcium hydroxide. na2co3 + ca(oh)2 → naoh + caco3 how many grams of naoh are produced from 20.0 grams of na2co3? (molar mass of na = 22.989 g/mol, c = 12.01 g/mol, o = 15.999 g/mol, ca = 40.078 g/mol, h = 1.008 g/mol) 12.2 grams 15.1 grams 24.4 grams 30.2 grams
Answers: 2

Chemistry, 22.06.2019 09:00
Achemist 16 drop copper metal from copper chloride solution. the chemist place is 0.50 g of aluminum foil in a solution containing 0.75 g of copper (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction?
Answers: 1

Chemistry, 22.06.2019 12:10
Consider the reaction: n2(g) + o2(g) ⇄ 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3
You know the right answer?
Questions

History, 07.07.2019 05:50


History, 07.07.2019 05:50

History, 07.07.2019 05:50


Chemistry, 07.07.2019 05:50


Biology, 07.07.2019 05:50

Social Studies, 07.07.2019 05:50

History, 07.07.2019 05:50

History, 07.07.2019 05:50

Biology, 07.07.2019 05:50

English, 07.07.2019 06:00

Social Studies, 07.07.2019 06:00


Business, 07.07.2019 06:00

English, 07.07.2019 06:00

History, 07.07.2019 06:00


Social Studies, 07.07.2019 06:00