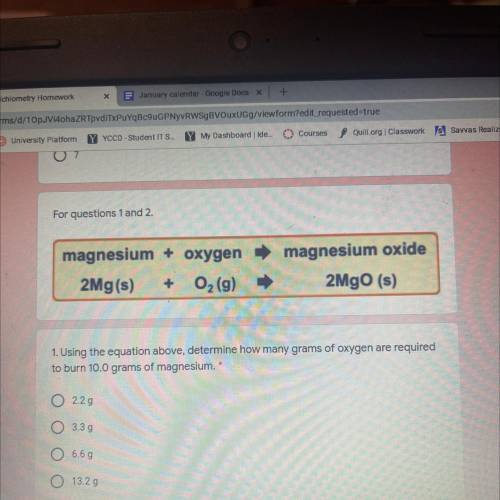
Magnesium + oxygen
2Mg(s) + O2 (9)
magnesium oxide
2MgO (s)
1. Using the equatio...

Chemistry, 11.02.2022 14:00 kayranicole1
Magnesium + oxygen
2Mg(s) + O2 (9)
magnesium oxide
2MgO (s)
1. Using the equation above, determine how many grams of oxygen are required
to burn 10.0 grams of magnesium.
2.29
O 3.39
6.69
Ο Ο
13.29

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 08:30
Joan writes four numbers on the board in standard form, and then she writes their scientific notation
Answers: 1

Chemistry, 22.06.2019 19:30
Estimate the molar mass of the gas that effuses at 1.6 times the effusion rate of carbon dioxide.
Answers: 1

Chemistry, 22.06.2019 20:00
How are the terms group and period used on the periodic table
Answers: 1

Chemistry, 22.06.2019 23:20
In medium-sized stars such as the sun, nuclear fusion almost always means the fusing of nuclei to form , but larger stars can produce elements as heavy as
Answers: 2
You know the right answer?
Questions


Chemistry, 06.09.2019 02:10

English, 06.09.2019 02:10


Computers and Technology, 06.09.2019 02:10


Chemistry, 06.09.2019 02:10

History, 06.09.2019 02:10












Computers and Technology, 06.09.2019 02:10



