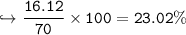Chemistry, 01.02.2022 21:50 annamerryberry1016
In this reaction: Mg (s) + I₂ (s) → MgI₂ (s), if 10.0 g of Mg reacts with 60.0 g of I₂, and 53.88 g of MgI₂ form, what is the percent yield?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 01:30
Idon't really understand this can you me and show your work.☺☺[ chemistry b] subject [ electron transfer in lonic bonds]grade( 12)
Answers: 1

Chemistry, 22.06.2019 07:00
If there is any 12 to 14 girls that need a boyfriend just follow me and let me know
Answers: 1

Chemistry, 22.06.2019 17:00
Which statement is true about a catalyst? a: a catalyst decreases the rate of the reaction. b. a catalyst is consumed during a chemical reaction. c. a catalyst lowers the activation energy of a reaction. d. a catalyst increases the reactant concentration during a reaction.
Answers: 1

You know the right answer?
In this reaction: Mg (s) + I₂ (s) → MgI₂ (s), if 10.0 g of Mg reacts with 60.0 g of I₂, and 53.88 g...
Questions

Mathematics, 21.11.2020 03:20

Physics, 21.11.2020 03:20

Mathematics, 21.11.2020 03:20

Mathematics, 21.11.2020 03:20

Mathematics, 21.11.2020 03:20

Biology, 21.11.2020 03:20

English, 21.11.2020 03:20



Computers and Technology, 21.11.2020 03:20




Health, 21.11.2020 03:20

Mathematics, 21.11.2020 03:20


Mathematics, 21.11.2020 03:20


Mathematics, 21.11.2020 03:20

Mathematics, 21.11.2020 03:20