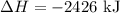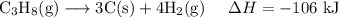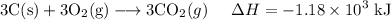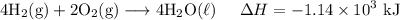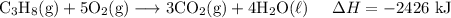H2 (g)+12O2 (g)→H2O (l) ΔH=−286 kJ

Chemistry, 18.01.2022 08:50 monkeyrose1999
Study the reactions.
C (s)+O2 (g)→CO2 (g) ΔH=−394 kJ
H2 (g)+12O2 (g)→H2O (l) ΔH=−286 kJ
3C (s)+4H2 (g)→C3H8 (g) ΔH=106 kJ
Target Reaction:
C3H8 (g)+5O2 (g)→3CO2 (g)+4H2O (l)ΔH= ?
What is the enthalpy change of the target reaction?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 18:00
How does climate change cause the ocean's thermohaline current to slow down?
Answers: 3

Chemistry, 22.06.2019 21:00
Which answer tells the reason the earth’s climate is getting warmer? too many animals are becoming extinct. large glaciers are melting in antarctica. the earth is moving closer to the sun. driving cars gives off gases that trap heat in the atmosphere.
Answers: 1

Chemistry, 23.06.2019 04:31
What are the coefficients that will balance the skeleton equation below? n2 + h2 → nh3
Answers: 1

Chemistry, 23.06.2019 05:30
Awhite powder is added to a solution. the images show observations made before the powder is added, just after the powder has been added, and a little while later. (the liquid in the small beaker is phenol red solution.) what evidence shows that a chemical change has taken place?
Answers: 1
You know the right answer?
Study the reactions.
C (s)+O2 (g)→CO2 (g) ΔH=−394 kJ
H2 (g)+12O2 (g)→H2O (l) ΔH=−286 kJ
H2 (g)+12O2 (g)→H2O (l) ΔH=−286 kJ
Questions







Mathematics, 30.08.2019 03:30

Spanish, 30.08.2019 03:30

Mathematics, 30.08.2019 03:30





Mathematics, 30.08.2019 03:30


Arts, 30.08.2019 03:30


Chemistry, 30.08.2019 03:30