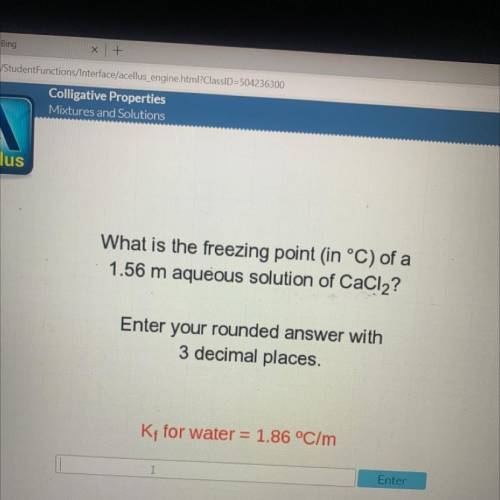
What is the freezing point in °C) of a
1.56 m aqueous solution of CaCl2?
Enter your rounded...

Chemistry, 02.01.2022 14:00 edimilperdomo
What is the freezing point in °C) of a
1.56 m aqueous solution of CaCl2?
Enter your rounded answer with
3 decimal places.
Kt for water = 1.86 °C/m

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 02:30
When svante arrhenius first proposed his acid-base theory, he was a doctoral candidate. his professors thought his ideas were unfounded. within a decade, the arrhenius theory of acid-base was widely accepted and praised within the scientific world. arrhenius defined acids as compounds having ionizable hydrogen and bases as compounds with ionizable a) barium. b) hydronium. c) hydroxide. d) oxygen.
Answers: 3



Chemistry, 22.06.2019 19:50
A2.5% (by mass) solution concentration signifies that there is a 2.5 % (by mass) solution concentration signifies that there is blank of solute in every 100 g of solution. of solute in every 100 g of solution
Answers: 3
You know the right answer?
Questions




Mathematics, 12.10.2021 03:10

History, 12.10.2021 03:10

Physics, 12.10.2021 03:10

Mathematics, 12.10.2021 03:10


Mathematics, 12.10.2021 03:10

Social Studies, 12.10.2021 03:10



Mathematics, 12.10.2021 03:10


History, 12.10.2021 03:10

English, 12.10.2021 03:10

Physics, 12.10.2021 03:10

Mathematics, 12.10.2021 03:10

Mathematics, 12.10.2021 03:10

Arts, 12.10.2021 03:20



