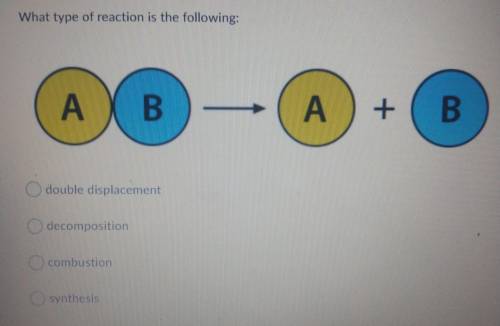
Please help need CORRECT answer
...

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 12:00
In a laboratory, 1.55mg of an organic compound containing carbon, hydrogen, and oxygen is burned for analysis. this combustion resulted in the formation of 1.45mg of carbon dioxide and .89 mg of water. what is the empirical formula for this compound?
Answers: 1

Chemistry, 23.06.2019 01:00
An unsaturated hydrocarbon is a hydrogen-carbon compound with a. a network solid structure b. single bonds c. single bonds in a branched-chain structure d. double or triple bonds
Answers: 1

Chemistry, 23.06.2019 01:30
Magnesium is the limiting reactant in this experiment. calculate the theoretical yield of mgo for each trial. trial 1: trial 2: data mass of empty crucible with lid trial 1: 26.688 trial 2: 26.681 mass of mg metal, crucible, and lid trial 1: 26.994 trial: 2 26.985 mass of mgo, crucible, and lid trial 1: 27.188 trial 2: 27.180
Answers: 1
You know the right answer?
Questions

Mathematics, 06.01.2020 23:31



Computers and Technology, 06.01.2020 23:31



Computers and Technology, 06.01.2020 23:31


Mathematics, 06.01.2020 23:31

Mathematics, 06.01.2020 23:31

Physics, 06.01.2020 23:31

Mathematics, 06.01.2020 23:31


Mathematics, 06.01.2020 23:31

Mathematics, 06.01.2020 23:31

Mathematics, 06.01.2020 23:31



Spanish, 06.01.2020 23:31