
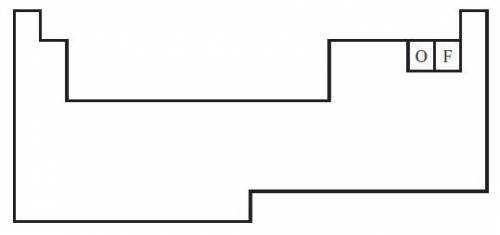
The diagram below shows a partial periodic table.
The electron configuration of oxygen is 1s2 2s2 2p4. On the periodic table, fluorine is one space to the right of oxygen. Which of the following electron configurations represents fluorine?
1s2 2s2 2p3
1s2 2s2 2p6 1s2 3s2 3p3
1s2 2s2 2p5

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 04:10
Answer from each drop-down menu. e characteristics of a borane molecule (bh). the lewis structure and table of electronegativities are given olecular shape is and the molecule is reset next erved. search e a
Answers: 2

Chemistry, 22.06.2019 05:50
In an exothermic reaction the bonding energy of the product is: less than the reactants same as the reactants greater than the reactants dependent upon the presence of a catalyst
Answers: 1

Chemistry, 22.06.2019 15:40
Use the periodic table to complete this equation that represents nuclear fission processesun - ba c 3 n
Answers: 2

You know the right answer?
The diagram below shows a partial periodic table.
The electron configuration of oxygen is 1s2 2s2...
Questions



Advanced Placement (AP), 02.12.2021 04:30


Mathematics, 02.12.2021 04:30

Mathematics, 02.12.2021 04:30

Mathematics, 02.12.2021 04:30


Mathematics, 02.12.2021 04:30



History, 02.12.2021 04:30

Mathematics, 02.12.2021 04:30


Social Studies, 02.12.2021 04:30


Business, 02.12.2021 04:30



Computers and Technology, 02.12.2021 04:30



