
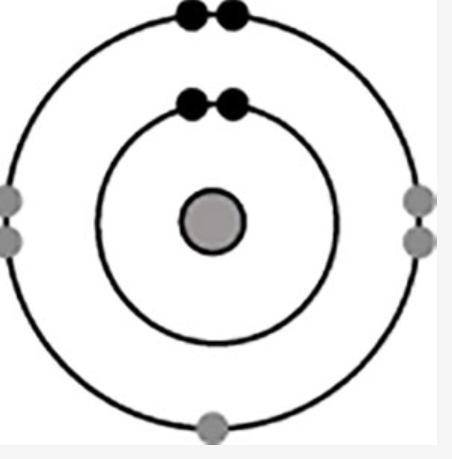
The diagram shows the electron configuration of an atom of an element for the electrons in the s and p orbitals.
A shaded circle is shown. There are two concentric circles around this shaded circle. In inner concentric circle has two dark shaded dots. The outer concentric circle has two dark shaded dots in the corresponding position to the inner concentric circle. The outer concentric circle also has five light shaded dots, two on the left and two on the right boundary and one at the bottom.
What is the group number of the element in the periodic table?
17
16
2
1

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 13:00
What happens to the average kinetic energy of a gas when the particles of the gas collide against each other at a constant temperature and volume? explain your answer.
Answers: 3

Chemistry, 22.06.2019 16:00
Click the button that shows the correct relationship of the electron affinities of the elements sodium and phosphorus. sodium’s electron affinity value is more negative than the electron affinity value of phosphorus. phosphorus’ electron affinity value is more negative than the electron affinity value of sodium. this information cannot be determined using the periodic table. answer is b on e2020.
Answers: 3

Chemistry, 22.06.2019 19:00
What information does a complete ionic equation give that the balanced equation doesn’t show?
Answers: 1

Chemistry, 23.06.2019 00:00
2-bromo-2-methylbutane undergoes an e1 elimination reaction in the presence of ethanol. in the next reaction only one of the possible products is represented. although the product shown is not the major product of the reaction, notice that there is more than one way it can be produced. complete the mechanism and draw the missing substances.
Answers: 1
You know the right answer?
The diagram shows the electron configuration of an atom of an element for the electrons in the s and...
Questions

Mathematics, 02.08.2019 23:00


Physics, 02.08.2019 23:00



Health, 02.08.2019 23:00

Social Studies, 02.08.2019 23:00

Advanced Placement (AP), 02.08.2019 23:00



History, 02.08.2019 23:00



History, 02.08.2019 23:00


English, 02.08.2019 23:00




Geography, 02.08.2019 23:00



