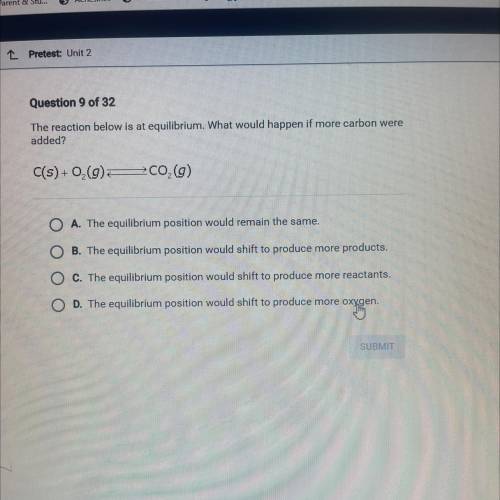
Question 9 of 32
The reaction below is at equilibrium. What would happen if more carbon were
...

Question 9 of 32
The reaction below is at equilibrium. What would happen if more carbon were
added?
C(s) +0,0 200,0)
A. The equilibrium position would remain the same.
B The equilibrium position would shift to produce more products.
The equilibrium position would shift to produce more reactants.
The equilibrium position would shift to produce more oxygen
SUNT

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 20:20
Calculate the molarity of the solution. 6.02 x 1022 molecules of hci (molecular weight = 36.5 g/mole) in 2.0 liters of water m
Answers: 1

Chemistry, 22.06.2019 07:00
In the cathode ray tube experiment, j. j. thomson passed an electric current through different gases inside a cathode ray tube in the presence of an electric field. in which two ways did this experiment change scientists’ understanding of the atom?
Answers: 2


Chemistry, 22.06.2019 10:00
Part 1: include important facts found through your research. part 2: include your visual display. include your summary of “the chemistry of water” from the national science foundation website. include your experiment. part 3: include responses to the reflection questions.
Answers: 1
You know the right answer?
Questions

English, 07.11.2019 23:31




Biology, 07.11.2019 23:31

English, 07.11.2019 23:31



Mathematics, 07.11.2019 23:31



History, 07.11.2019 23:31

Mathematics, 07.11.2019 23:31

Mathematics, 07.11.2019 23:31




History, 07.11.2019 23:31


Mathematics, 07.11.2019 23:31



