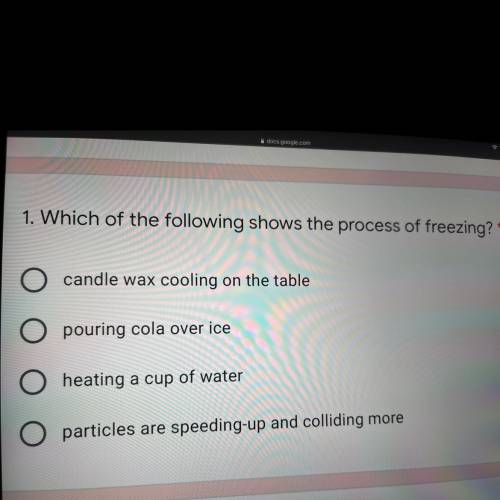
1. Which of the following shows the process of freezing?'
A. candle wax cooling on the table
...

Chemistry, 09.12.2021 21:00 veronicatrejoaguiler
1. Which of the following shows the process of freezing?'
A. candle wax cooling on the table
B. pouring cola over ice
C. heating a cup of water
D. particles are speeding-up and colliding more

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 03:30
The atomic radius of sodium is 186 pm and of chlorine is 100 pm. the ionic radius for na+ is 102 pm and for cl– is 181 pm. in going from na to cl in period 3, why does the atomic radius decrease while the ionic radius increases? a. the inner electrons in the sodium cation shield its valence electrons more effectively than the inner electrons in the chloride anion do. b. the inner electrons shield the valence electrons more effectively in the chlorine atom than in the chloride anion. c. the outermost electrons in chloride experience a smaller effective nuclear charge than those in the sodium cation do. d. the outermost electrons in chloride experience a larger effective nuclear charge than those in the sodium cation do. e. monatomic ions are bigger than the atoms from which they are formed.
Answers: 2

Chemistry, 22.06.2019 17:40
Which statement about hf is true? it is zero for any compound in its standard state. it is positive when the bonds of the product store more energy than those of the reactants. it is negative when a compound forms from elements in their standard states. it is zero for any element that is in the liquid state.
Answers: 1

Chemistry, 22.06.2019 23:00
What extra step distinguishes fermentation from glycolysis
Answers: 1

You know the right answer?
Questions

History, 27.10.2019 23:43




History, 27.10.2019 23:43




Health, 27.10.2019 23:43


Mathematics, 27.10.2019 23:43



Mathematics, 27.10.2019 23:43



Health, 27.10.2019 23:43

History, 27.10.2019 23:43

History, 27.10.2019 23:43

English, 27.10.2019 23:43