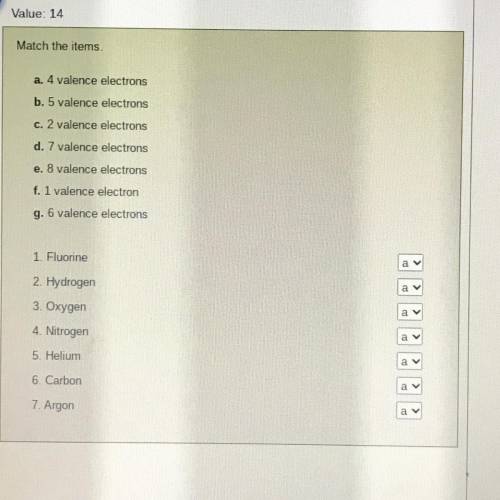
1. Fluorine - D. 7 valence electrons
Explanation :
For example, fluorine has seven valence electrons, so it is most likely to gain one electron to form an ion with a 1- charge
2. Hydrogen - F. 1 valence electrons
Explanation :
Hydrogen's valence number is one, because it has only one valence electron and needs only one shared electron to fill its energy levels. This means it can bond with many elements. For example, four hydrogen atoms can bond with a carbon atom, which has four valence electrons, to form methane.
3. Oxygen - G. 6 velance electrons
Explanation :
Valence electrons are the electrons in the outermost shell, or energy level, of an atom. For example, oxygen has six valence electrons, two in the 2s subshell and four in the 2p subshell. We can write the configuration of oxygen's valence electrons as 2s²2p⁴.
4. Nitrogen - B. 5 velance electrons
Explanation :
Nitrogen has a total of 5 valence electrons, so doubling that, we would have a total of 10 valence electrons with two nitrogen atoms. The octet requires an atom to have 8 total electrons in order to have a full valence shell, therefore it needs to have a triple bond.
5. Helium - C. 2 velance electrons
Explanation :
Helium has 2 electrons --- both in the first shell (so two valence electrons). Lithium has 3 electrons --- 2 in the first shell, and 1 in the second shell (so one valence electron).
6. Carbon - A. 4 velance electrons
Explanation :
All the carbon group atoms, having four valence electrons, form covalent bonds with nonmetal atoms; carbon and silicon cannot lose or gain electrons to form free ions, whereas germanium, tin, and lead do form metallic ions but only with two positive charges.
7. Argon - E. 8 velance electrons
Explantion :
So, the number of electrons in the Argon atom is 18 because it is present in the neutral state. Hence, from the above given configuration it is clear that the number of valence electrons in the Argon atom is eight.