
Chemistry, 03.12.2021 01:50 recon12759
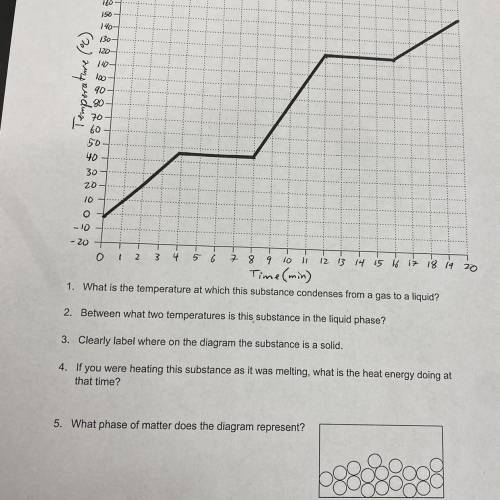
1. What is the temperature at which this substance condenses from a gas to a liquid?
2. Between what two temperatures is this substance in the liquid phase?
3. Clearly label where on the diagram the substance is a solid.
4. If you were heating this substance as it was melting, what is the heat energy doing at that time?
5. What phase of matter does the diagram represent?

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 17:30
How many grams of magnesium metal will react completely with 8.3 liters of 5.5 m hcl? show all of the work needed to solve this problem. mg (s) + 2hcl (aq) → mgcl2 (aq) + h2 (g)
Answers: 3

Chemistry, 22.06.2019 05:50
What are transitions between a liquid and a solid called? identify which way they are transitioning
Answers: 2

Chemistry, 22.06.2019 10:50
How many grams of oxygen gas are contained in a 15 l sample at 1.02 atm and 28°c? show your work.
Answers: 1

Chemistry, 22.06.2019 12:30
Place the elements below in order of decreasing ionization energy. aluminum(al) chlorine(cl) magnesium (mg) sulfur(s)
Answers: 1
You know the right answer?
1. What is the temperature at which this substance condenses from a gas to a liquid?
2. Between wh...
Questions

Mathematics, 12.07.2019 08:30




History, 12.07.2019 08:30


History, 12.07.2019 08:30

Mathematics, 12.07.2019 08:30


Chemistry, 12.07.2019 08:30


Business, 12.07.2019 08:30

Social Studies, 12.07.2019 08:30

Computers and Technology, 12.07.2019 08:30






Social Studies, 12.07.2019 08:30



