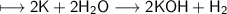Chemistry, 28.11.2021 09:50 sterlingrobinson35
When potassium metal is placed in water, a large amount of energy is released as potassium hydroxide and hydrogen gas are produced in the reaction 2K(s) + 2H2O(l) → 2KOH(aq) + H2(g). Your lab partner says this is a redox reaction and a combustion reaction. Do you agree? Defend your answer by explaining whether or not it meets the requirements of each type of reaction.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 15:00
If a reaction has g of -136kj at 110°c, will it be spontaneous at this temperature (110°c)? yes or no
Answers: 2

Chemistry, 22.06.2019 00:00
For ai it's atomic number is 13 and it's mass number is 27 how many neutrons does it have
Answers: 1

Chemistry, 22.06.2019 09:30
One way that radioactive waste is treated is by burying it in repositories. the repositories are found only in states with very low populations. true or false? a. trueb. false(also i meant to put high school but it put down middle school instead)
Answers: 1

You know the right answer?
When potassium metal is placed in water, a large amount of energy is released as potassium hydroxide...
Questions


Biology, 11.05.2021 21:00




Social Studies, 11.05.2021 21:00

Mathematics, 11.05.2021 21:00




Mathematics, 11.05.2021 21:00



Arts, 11.05.2021 21:00


Mathematics, 11.05.2021 21:00



Mathematics, 11.05.2021 21:00