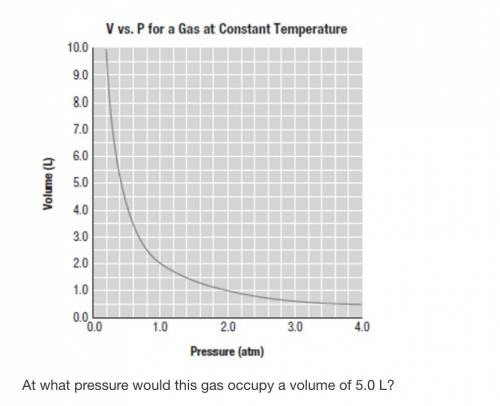
At what pressure does the gas occupy a volume of 5 L? What is the volume of the gas at a pressure of 5 atm? Calculate the Boyle's law constant at a volume of 2 L. Calculate the Boyle's law constant at a pressure of 2 atm. Calculate the pressure, in atm, required to compress a sample of helium gas from 20.9 L (at 1.00 atm) to 4.00L. A balloon filled with helium gas at 1.00 atm occupies 15.6 L. What volume would the balloon occupy in the upper atmosphere at a pressure of 0.150 atm? State Charles's law in words. State Charles's law in equation form. Explain why the Kelvin scale is used for gas law calculations. The temperature on a summer day may be 90 degree F. Convert this value to Kelvin units. The temperature of a gas is raised from 25 degree C to 50 degree C Will the volume double if mass and pressure do not change? Why or why not? Verify your answer to Question 5.45 by calculating the temperature needed to double the volume of the gas. Determine the change in volume that takes place when a 2.00-L sample of N_2 (g) is heated from 250 degree C to 500 degree C. Determine the change in volume that takes place when a 2.00-L sample of N_2 (g) is heated from 250 degree K to 500 degree K. A balloon containing a sample of helium gas is warmed in an oven. If the balloon measures 1.25 L at room temperature (20 degree C), what is its volume at 80 degree C? The balloon described in Question 5.49 was then placed in a refrigerator at 39 degree F. Calculate its new volume. A balloon, filled with N_2, has a volume of 2.00 L at an indoor temperature of 68 degree F. When placed outdoors, the volume was observed to increase to 2.20 L. What is the outdoor temperature in degree F? A balloon, filled with an ideal gas, has a volume of 5.00 L at what temperature (degree F) would the balloon's volume double?