
Chemistry, 24.11.2021 01:50 joycetleiji1
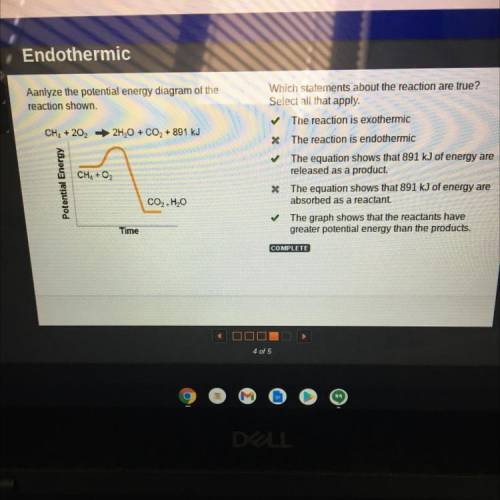
Aanlyze the potential energy diagram of the
reaction shown.
Which statements about the reaction are true?
Select all that apply.
The reaction is exothermic
The reaction is endothermic
The equation shows that 891 kJ of energy are
released as a product.
The equation shows that 891 kJ of energy are
absorbed as a reactant.
The graph shows that the reactants have greater potential energy than the products.
The answer is a, c,e

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 04:00
Acontainer holds 35.8 moles of gas under 10.0 atm of pressure at 70.0 c. what is the volume of the container?
Answers: 2

Chemistry, 22.06.2019 07:30
Label a-f based on the table using c for concentrated and d for dilute
Answers: 2

Chemistry, 22.06.2019 11:40
Calculate the number of kilojoules to warm 125 g of iron from 23.5°c to 78.0°c.
Answers: 3

Chemistry, 22.06.2019 14:00
What is the ph of a solution that has a hydrogen ion concentration of 1.0 * 10 -9 m?
Answers: 3
You know the right answer?
Aanlyze the potential energy diagram of the
reaction shown.
Which statements about the react...
Which statements about the react...
Questions



Mathematics, 03.01.2020 05:31


Biology, 03.01.2020 05:31

World Languages, 03.01.2020 05:31


History, 03.01.2020 05:31





History, 03.01.2020 05:31

Health, 03.01.2020 05:31



Chemistry, 03.01.2020 05:31





