
Chemistry, 26.10.2021 22:20 cordobamariana07
Please help me with this ill mark brainliest please please im literally crying here
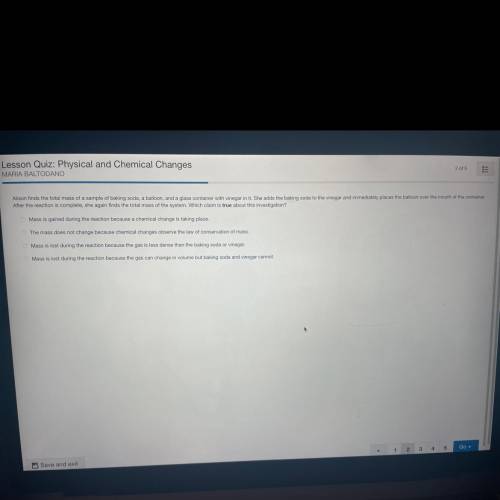

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 06:30
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 3


Chemistry, 22.06.2019 14:50
Given the following information: mass of proton = 1.00728 amu mass of neutron = 1.00866 amu mass of electron = 5.486 × 10^-4 amu speed of light = 2.9979 × 10^8 m/s calculate the nuclear binding energy (absolute value) of 3li^6. which has an atomic mass of 6.015126 amu. j/mol.
Answers: 2
You know the right answer?
Please help me with this ill mark brainliest please please im literally crying here
...
...
Questions

History, 01.09.2019 01:30



English, 01.09.2019 01:30



Mathematics, 01.09.2019 01:30

Social Studies, 01.09.2019 01:30

Computers and Technology, 01.09.2019 01:30




Physics, 01.09.2019 01:30

English, 01.09.2019 01:30

Physics, 01.09.2019 01:30

Mathematics, 01.09.2019 01:30

English, 01.09.2019 01:30



English, 01.09.2019 01:30



