Please help with the question attached
I need it in an hour
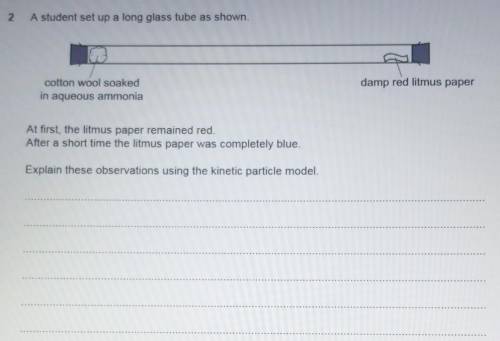
...

Chemistry, 22.10.2021 22:30 itscheesycheedar
Please help with the question attached
I need it in an hour

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 08:00
Why is the bond angle in a water molecule less than the bond angle of methane? a. the central oxygen atom in water has two lone pairs of electrons, whereas the central carbon atom in methane has no lone pairs. b. the central hydrogen atom in water has one lone pair of electrons, whereas the central carbon atom in methane has two lone pairs. c. the central oxygen atom in water has four lone pairs of electrons, whereas the central carbon atom in methane has only one lone pair. d. the central oxygen atom exerts more repulsive force on surrounding atoms than the central carbon atom in methane does. reset next
Answers: 2

Chemistry, 22.06.2019 08:30
Identify one disadvantage to each of the following models of electron configuration: -dot structures -arrow and line diagrams -written electron configurations type in your answer below. (answer) -dot structures do not show the distribution of electrons in orbitals and take up a lot of space. -arrow and line diagrams take up a lot of space and make it difficult to count electrons. -written configurations make it easy to lose count of electrons and do not show the distribution of electrons in orbitals.
Answers: 3


You know the right answer?
Questions




Physics, 21.03.2020 22:12

Mathematics, 21.03.2020 22:12

History, 21.03.2020 22:12

Mathematics, 21.03.2020 22:12


Mathematics, 21.03.2020 22:12

Chemistry, 21.03.2020 22:12


Mathematics, 21.03.2020 22:12

English, 21.03.2020 22:12



English, 21.03.2020 22:13


History, 21.03.2020 22:13




