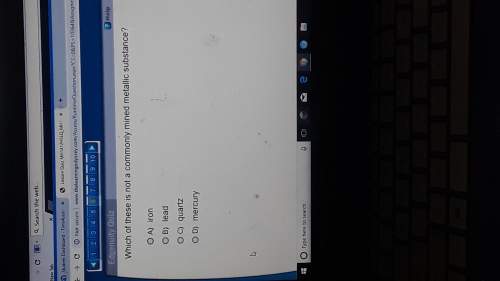
Chemistry, 22.10.2021 21:20 krystal408
. Why does the element with the higher atomic number have a lower ionization energy than the element with the lower atomic number?

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 19:10
Which statement correctly describes the phosphate ion, ? it is composed of one phosphorus atom and four oxygen atoms covalently bonded together, and there is a –3 charge distributed over the entire ion. it is composed of one phosphorus atom and four oxygen atoms covalently bonded together, and there is a –3 charge on the phosphorus atom. it is composed of one phosphorus atom and four oxygen atoms ionically bonded together, and there is a –3 charge distributed over the entire ion. it is composed of one phosphorus atom and four oxygen atoms ionically bonded together, and there is a –3 charge on the phosphorus atom.
Answers: 3


Chemistry, 22.06.2019 23:00
Movement that is like a t a type of wave that transfers energy where the particles in the medium move in a circle motion while the energy travels left or right. a type of wave that transfers energy where the particles in the medium move perpendicular to the direction in which the energy is traveling. transfers energy from one location to another a type of wave that transfers energy where the particles in the medium move parallel to the direction in which the energy is traveling. movement that is back and forth, like an equal sign = 1. wave 2. parallel movement 3. perpendicular movement 4. transverse wave 5. longitudinal wave 6. surface wave
Answers: 1
You know the right answer?
. Why does the element with the higher atomic number have a lower ionization energy than the element...
Questions

Mathematics, 16.12.2019 01:31

Arts, 16.12.2019 01:31

Mathematics, 16.12.2019 01:31





Mathematics, 16.12.2019 01:31




Social Studies, 16.12.2019 01:31


Mathematics, 16.12.2019 01:31





Chemistry, 16.12.2019 01:31

History, 16.12.2019 01:31