X and Y form a covalent compound.

Chemistry, 10.10.2021 14:00 ThereaalistNy
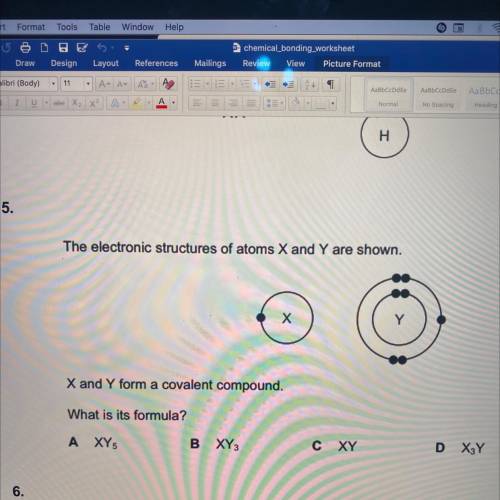
The electronic structures of atoms X and Y are shown.
х
X and Y form a covalent compound.
What is its formula?
A XY
B XY
C
XY
D XY

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 14:50
Given the following information: mass of proton = 1.00728 amu mass of neutron = 1.00866 amu mass of electron = 5.486 × 10^-4 amu speed of light = 2.9979 × 10^8 m/s calculate the nuclear binding energy (absolute value) of 3li^6. which has an atomic mass of 6.015126 amu. j/mol.
Answers: 2

Chemistry, 23.06.2019 00:00
The empirical formula of a compound is ch2o and its mass is 120 amu/molecule, what is its formula?
Answers: 2

Chemistry, 23.06.2019 03:30
The semi-conductors on the periodic table are classified as
Answers: 1

Chemistry, 23.06.2019 04:31
2ki + pb(no3)2 → 2kno3 + pbi2 determine how many moles of kno3 are created if 0.03 moles of ki are completely consumed.
Answers: 1
You know the right answer?
The electronic structures of atoms X and Y are shown.
х
X and Y form a covalent compound.
X and Y form a covalent compound.
Questions

Mathematics, 12.12.2020 16:10

Health, 12.12.2020 16:10

Mathematics, 12.12.2020 16:10



Mathematics, 12.12.2020 16:10

Mathematics, 12.12.2020 16:10

English, 12.12.2020 16:10

Mathematics, 12.12.2020 16:10






History, 12.12.2020 16:10

Mathematics, 12.12.2020 16:10



Mathematics, 12.12.2020 16:10

Chemistry, 12.12.2020 16:10



