Hello there im looking for some help here thanks!
ill give brainliest if im able to!
...

Chemistry, 07.10.2021 14:00 justhereforanswers13
Hello there im looking for some help here thanks!
ill give brainliest if im able to!
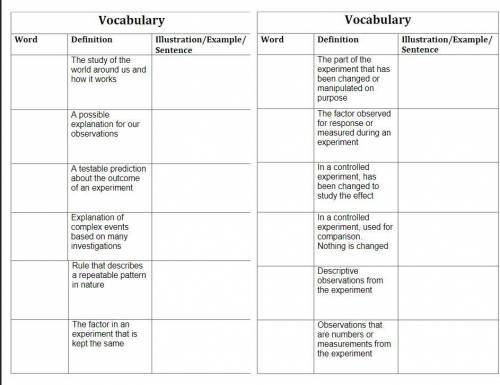
Use this list of words:
1. science
2. inference
3. hypothesis
4. scientific theory
5. scientific law
6. constant
7. independent variable
8. dependent variable
9. experimental group
10. control group
11. qualitative data
12. quantitative data

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 21:00
Match the following items. 1. high-intensity bundle of energy being emitted from some decaying nuclei gamma ray 2. particle radiating from the nucleus of some atoms beta particle 3. negative particle identical to an electron but radiating from a decaying nucleus alpha particle
Answers: 1

Chemistry, 22.06.2019 14:30
Consider the reduction reactions and their equilibrium constants. cu+(aq)+e−↽−−⇀cu(s)pb2+(aq)+2e−↽−−⇀pb(s)fe3+(aq)+3e−↽−−⇀fe(=6.2×108=4.0×10−5=9.3×10−3 cu + ( aq ) + e − ↽ − − ⇀ cu ( s ) k =6.2× 10 8 pb 2 + ( aq ) +2 e − ↽ − − ⇀ pb ( s ) k =4.0× 10 − 5 fe 3 + ( aq ) +3 e − ↽ − − ⇀ fe ( s ) k =9.3× 10 − 3 arrange these ions from strongest to weakest oxidizing agent.
Answers: 3

Chemistry, 22.06.2019 21:00
Kp is the equilibrium constant for dissociation of the propionic acid dimer. what is the sign of the slope for a plot of the natural logarithm of kp vs. inverse temperature for this reaction?
Answers: 1

Chemistry, 22.06.2019 21:30
Plzz a sample of table sugar (sucrose, c12h22o11) has a mass of 7.801 g. ● a) calculate the number of moles of c12h22o11 in the sample b) calculate the number of moles of each element in c12h22o11 (number of moles of c, number of moles of h & number of moles of o) in the sample. (use your answer from part a as your starting point.) show your work and highlight your final answer. calculate the number of atoms of each element in c12h22o11 (number of atoms of c, number of atoms of h & number of atoms of o) in the sample. (use your answers from part b as your starting for each element.) show your work and highlight your final answer.
Answers: 1
You know the right answer?
Questions


Mathematics, 23.06.2021 05:50

History, 23.06.2021 05:50

Mathematics, 23.06.2021 05:50





World Languages, 23.06.2021 05:50



Chemistry, 23.06.2021 05:50

English, 23.06.2021 05:50

Mathematics, 23.06.2021 05:50

Mathematics, 23.06.2021 05:50


Chemistry, 23.06.2021 05:50

Arts, 23.06.2021 05:50

Mathematics, 23.06.2021 05:50

English, 23.06.2021 05:50



