
Chemistry, 06.10.2021 22:20 tatertottheyoungin
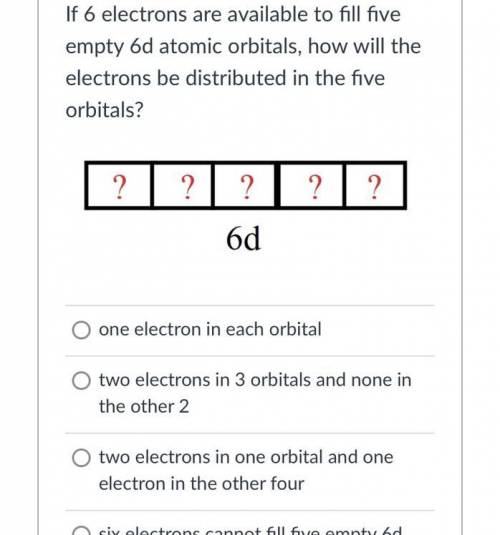
If 6 electrons are available to fill fiveempty 6d atomic orbitals, how will theelectrons be distributed in the fiveorbitals?? ? ? 2 ?6dO one electron in each orbitalO two electrons in 3 orbitals and none inthe other 2two electrons in one orbital and oneelectron in the other fourO six electrons cannot fill five empty 6datomic orbitals

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 07:50
In which situation can a mixture always be called a solution
Answers: 3

Chemistry, 22.06.2019 12:00
What is the percentage of hydrogen in nitrogen trihydride
Answers: 1


Chemistry, 22.06.2019 13:00
What is the mass of 2.00 l of an intravenous glucose solution with a density of 1.15 g/ml?
Answers: 2
You know the right answer?
If 6 electrons are available to fill fiveempty 6d atomic orbitals, how will theelectrons be distribu...
Questions

English, 20.02.2020 03:43

Mathematics, 20.02.2020 03:43


Computers and Technology, 20.02.2020 03:43




Computers and Technology, 20.02.2020 03:43

Mathematics, 20.02.2020 03:43


Chemistry, 20.02.2020 03:43


Mathematics, 20.02.2020 03:43


Mathematics, 20.02.2020 03:43

Social Studies, 20.02.2020 03:43

Social Studies, 20.02.2020 03:43


Biology, 20.02.2020 03:43

Biology, 20.02.2020 03:43



