
Chemistry, 06.10.2021 21:20 davidswafforddd478
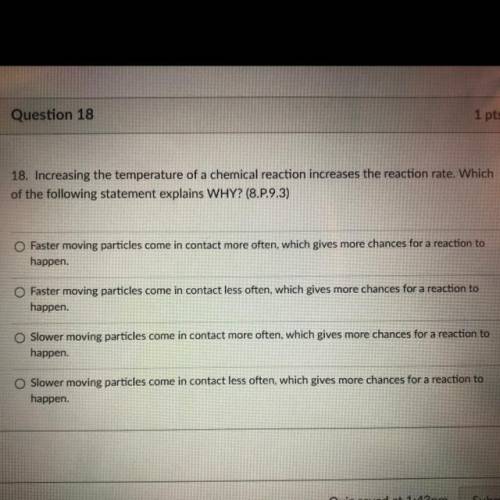
18. Increasing the temperature of a chemical reaction increases the reaction rate. Which
of the following statement explains WHY? (8.P.9.3)
O Faster moving particles come in contact more often, which gives more chances for a reaction to
happen.
Faster moving particles come in contact less often, which gives more chances for a reaction to
happen
Slower moving particles come in contact more often, which gives more chances for a reaction to
happen.
Slower moving particles come in contact less often, which gives more chances for a reaction to
happen.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 09:00
Given the following reaction: c3h8+5o2=3co2+4h20 how many grams of co2 will be produced 7 g of c3h8 and 98 g of o2
Answers: 1

Chemistry, 22.06.2019 14:00
The content of manganese (mn) in steel was determined spectrophotometrically and with the use of the standard addition method. an unknown sample of mn from a digested steel sample gave an absorbance of 0.185 when analyzed spectrophotometrically. when 5.00 ml of solution containing 95.5 ppm mn was added to 50.0 ml of the unknown steel solution (digested sample), the absorbance was 0.248. calculate the concentration, in parts-per-million (ppm), of mn in the digested steel sample solution.
Answers: 3

Chemistry, 22.06.2019 14:00
What is the ph of a solution that has a hydrogen ion concentration of 1.0 * 10 -9 m?
Answers: 3

Chemistry, 22.06.2019 18:00
Chlorophyll a had the molecular formula c55h72mgn4o5 how many atoms are in this molecule
Answers: 2
You know the right answer?
18. Increasing the temperature of a chemical reaction increases the reaction rate. Which
of the fo...
Questions

Mathematics, 07.12.2020 15:20


Social Studies, 07.12.2020 15:20

Mathematics, 07.12.2020 15:20

Computers and Technology, 07.12.2020 15:20

World Languages, 07.12.2020 15:20



Mathematics, 07.12.2020 15:20

Geography, 07.12.2020 15:20

Arts, 07.12.2020 15:20

Spanish, 07.12.2020 15:20

Advanced Placement (AP), 07.12.2020 15:20


Medicine, 07.12.2020 15:20

Mathematics, 07.12.2020 15:20

Mathematics, 07.12.2020 15:20

Chemistry, 07.12.2020 15:20


History, 07.12.2020 15:20



