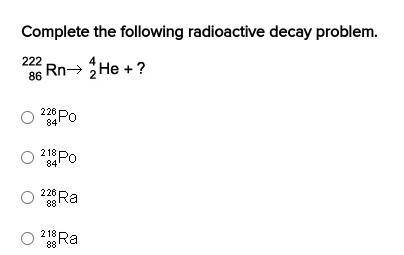I Need Help Fast Please
...

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 18:00
In a sample of oxygen gas at room temperature, the average kinetic energy of all the balls stays constant. which postulate of kinetic molecular theory best explains how this is possible? a. attractive forces between gas particles are negligible because the particles of an ideal gas are moving so quickly. b. collisions between gas particles are elastic; there is no net gain or loss of kinetic energy. c. gases consist of a large number of small particles, with a lot of space between the particles. d. gas particles are in constant, random motion, and higher kinetic energy means faster movement.
Answers: 1

Chemistry, 22.06.2019 12:00
Ican determine the molar mass of an element by looking on the under the atomic mass for the element. for example the molar mass of phosphorus is 30.974 grams/mole. avogadro’s number tells me the amount of representative particles in 1 mole of any substance. this means 12.011 gram sample of carbon and a 32.0 gram sample of sulfur have the same number of atoms.
Answers: 1

Chemistry, 22.06.2019 17:10
Increasing the substrate concentration in an enzymatic reaction could overcome which of the following? a) the need for a coenzymeb) allosteric inhibitionc) competitive inhibitiond) insufficient cofactors
Answers: 1

Chemistry, 22.06.2019 21:00
One similarity and one difference between an element and a mixture of elements
Answers: 1
You know the right answer?
Questions



Biology, 02.05.2021 14:00


Mathematics, 02.05.2021 14:00


Physics, 02.05.2021 14:00


Mathematics, 02.05.2021 14:00


Mathematics, 02.05.2021 14:00


English, 02.05.2021 14:00

Mathematics, 02.05.2021 14:00


Physics, 02.05.2021 14:00

Physics, 02.05.2021 14:00

Mathematics, 02.05.2021 14:00

Business, 02.05.2021 14:00

Mathematics, 02.05.2021 14:00