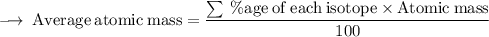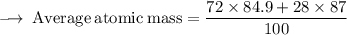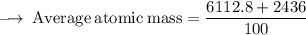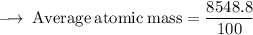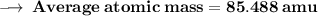Chemistry, 22.09.2021 14:00 demetricejames
Element X has two isotopes. If 72.0% of the element has an isotope mass of 84.9 atomic mass units, and 28.0% of the element has an isotopic mass of 87.0 atomic mass units, the average atomic mass of element X is numerically equal to

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 02:40
Consider the nuclear equation below. 239/94 pu—-> x+ 4/2 he. what is x?
Answers: 2

Chemistry, 22.06.2019 08:30
Which of the following would have less momentum than a 52 kg cheetah running at 10 m/s?
Answers: 2

Chemistry, 23.06.2019 00:00
What conclusion can you draw from this experiment about the components of the black ink?
Answers: 3

Chemistry, 23.06.2019 01:30
What happens to the concentration of hydronium ions as the ph of a solution increases? a. hydronium ion concentration stays the same b. hydronium ion concentration decreases c. hydronium ion concentration increases
Answers: 1
You know the right answer?
Element X has two isotopes. If 72.0% of the element has an isotope mass of 84.9 atomic mass units, a...
Questions

Mathematics, 22.05.2021 08:00




Chemistry, 22.05.2021 08:00




Mathematics, 22.05.2021 08:00

Biology, 22.05.2021 08:00


English, 22.05.2021 08:00

Mathematics, 22.05.2021 08:00



English, 22.05.2021 08:00


Mathematics, 22.05.2021 08:00