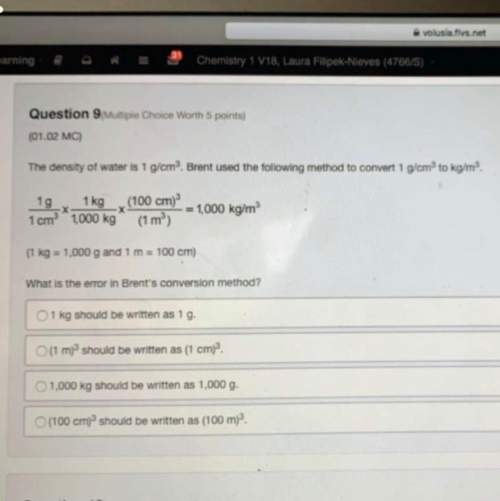
Balance this equation step by step.
HN03 + S >H2SO4+ NO
To identify which element i...

Chemistry, 19.09.2021 09:20 jeffhuffle17
Balance this equation step by step.
HN03 + S >H2SO4+ NO
To identify which element is oxidized and which element is reduced, you must assign numbers to all elements.
The element that increases in oxidation number is while the element.
that decreases in oxidation number is

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 23:00
At room temperature what happens to the average kinetic energy of the molecules of a solid, liquid, and a gas
Answers: 2

Chemistry, 22.06.2019 04:20
Which formula can be used to calculate the molar mass of ammonia (nh3)? molar mass of n + molar mass of h 3 × molar mass of n + molar mass of h molar mass of n + 3 × molar mass of h 3 × molar mass of n + 3 × molar mass of h
Answers: 1

Chemistry, 22.06.2019 19:30
What is the common name for the compound shown here? enter the common name of the compound shown?
Answers: 2

Chemistry, 22.06.2019 21:00
Which of the following is a physical property flammability heat of combustion solubility and toxicity
Answers: 1
You know the right answer?
Questions



History, 05.05.2020 16:21

Mathematics, 05.05.2020 16:21





Biology, 05.05.2020 16:21




Physics, 05.05.2020 16:21







English, 05.05.2020 16:21