Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 15:00
Which element in the third period would you expect to have the larger atomic radius, sodium (na) or sulfur (s)? a. sodium, because it has a higher effective nuclear charge attracting electrons in fewer energy levels. b. sodium, because it has fewer protons attracting electrons in the same energy levels. c. sulfur, because it has more protons attracting electrons in more energy levels. d. sulfur, because it has a higher effective nuclear charge attracting electrons in the same energy levels.
Answers: 2

Chemistry, 21.06.2019 23:00
Matches the chemical name of each oxide of phosphorus to its chemical formula
Answers: 2

Chemistry, 22.06.2019 02:20
Compared with the freezing-point depression of a 0.01 m c6h12o6 solution, the freezing-point depression of a 0.01 m nacl solution is
Answers: 1

You know the right answer?
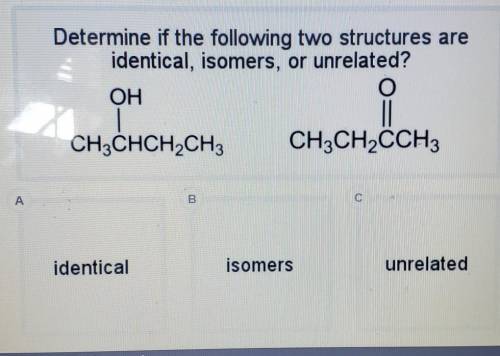
Determine if the following two structures are identical, isomers, or unrelated? OH O CH3CHCH2CH3 CH3...
Questions

Health, 03.06.2020 13:23

English, 03.06.2020 13:23



Mathematics, 03.06.2020 13:23

Mathematics, 03.06.2020 13:23

Social Studies, 03.06.2020 13:23



Mathematics, 03.06.2020 13:23

Mathematics, 03.06.2020 13:23

English, 03.06.2020 13:23

Mathematics, 03.06.2020 13:23

Mathematics, 03.06.2020 13:23

Mathematics, 03.06.2020 13:23




Mathematics, 03.06.2020 13:23