Chemistry, 12.09.2021 16:20 Dragonskeld
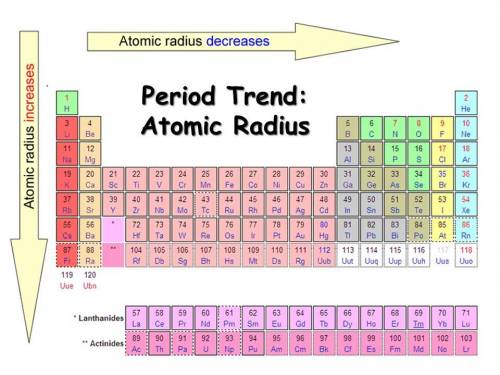
Arrange the following elements in order of increasing from smallest to largest atomic size K, O, Cs, Se explain why the reason why the trend exist.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 09:50
Although respiratory organs vary across different organisms, they all contain respiratory surfaces that have a large surface area and are extremely thin. explain why having an extremely thin respiratory surface with a large surface area is advantageous for the process of gas exchange
Answers: 1

Chemistry, 22.06.2019 12:30
According to the valence shell electron pair repulsion (vsepr) theory, a molecule that has four electron groups around the central atom will exhibit what electron geometry? view available hint(s) according to the valence shell electron pair repulsion (vsepr) theory, a molecule that has four electron groups around the central atom will exhibit what electron geometry? trigonal bipyramidal tetrahedral square planar determination of electron geometry requires information on whether the electron groups are lone pairs or bonding groups.
Answers: 2

Chemistry, 22.06.2019 13:30
How many protons, electrons, and neutrons are in each of the following isotopes? a. zirconium-90 b. palladium-108 c. bromine-81 d. antimony-123
Answers: 1

Chemistry, 22.06.2019 14:00
In the space, show a correct numerical setup for calculating the number of moles of co2 present in 11 grams of co2
Answers: 1
You know the right answer?
Arrange the following elements in order of increasing from smallest to largest atomic size K, O, Cs,...
Questions






World Languages, 17.07.2019 08:00


Advanced Placement (AP), 17.07.2019 08:00


History, 17.07.2019 08:00

Mathematics, 17.07.2019 08:00

Mathematics, 17.07.2019 08:00



English, 17.07.2019 08:00


Health, 17.07.2019 08:00