The substance that releases 21.2 kJ of energy when 1.42 mol of it freezes is_
...

Chemistry, 07.09.2021 20:30 phillswift2020
The substance that releases 21.2 kJ of energy when 1.42 mol of it freezes is_
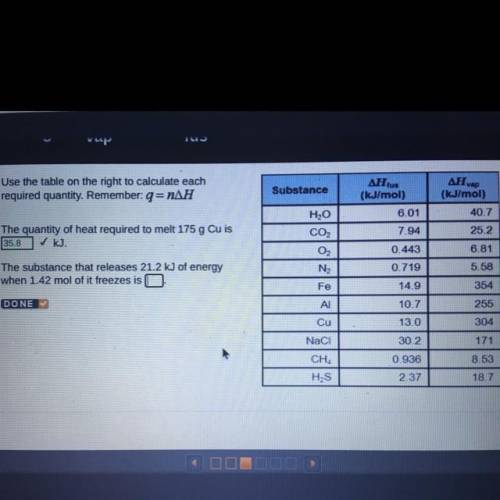

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 19:20
The equation picture below shows which type of nuclear reaction u 235 + n x e 134 + sr 100 + 2n
Answers: 1

Chemistry, 23.06.2019 01:00
Which substance—wood or silver—is the better thermal conductor? a thermal conductor is a material that requires very little heat energy to change its temperature. explain your answer.
Answers: 3

Chemistry, 23.06.2019 09:00
A0.10 m aqueous solution of sodium sulfate is a better conductor of electricity than a 0.10 m aqueous solution of sodium chloride. which of the following best explains this observation? (a) sodium sulfate is more soluble in water than sodium chloride. (b) sodium sulfate has a higher molar mass than sodium chloride. (c) to prepare a given volume of 0.10 m solution, the mass of sodium sulfate needed is more than twice the mass of sodium chloride needed. (d) more moles of ions are present in a given volume of 0.10 m sodium sulfate than in the same volume of 0.10 m sodium chloride. (e) the degree of dissociation of sodium sulfate in solution is significantly greater than that of sodium chloride.
Answers: 2
You know the right answer?
Questions


Mathematics, 20.09.2019 06:30





Mathematics, 20.09.2019 06:30

Biology, 20.09.2019 06:30

Health, 20.09.2019 06:30

Mathematics, 20.09.2019 06:30

Social Studies, 20.09.2019 06:30

History, 20.09.2019 06:30


Mathematics, 20.09.2019 06:30


Mathematics, 20.09.2019 06:30


English, 20.09.2019 06:30

Mathematics, 20.09.2019 06:30

Mathematics, 20.09.2019 06:30



