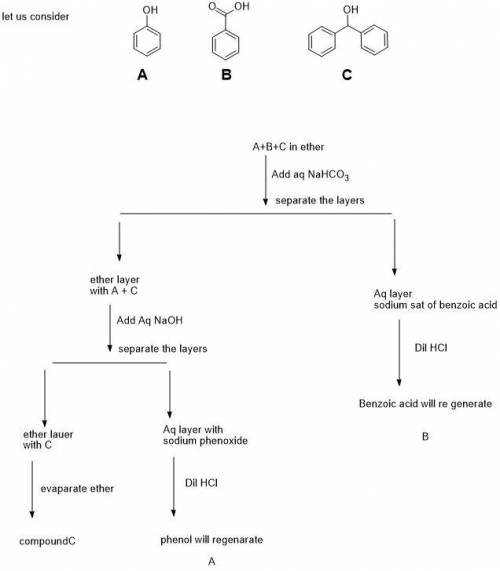
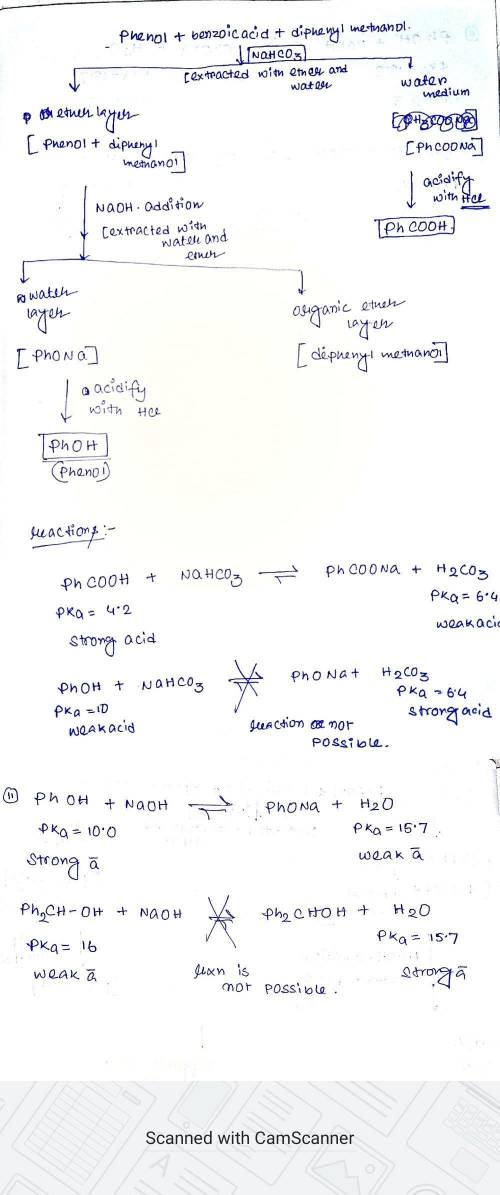
Createa flow chart separating phenol (P, pKa= 10.0), benzoic acid (B, pKa= 4.2), and diphenylmethanol (D, pKa~ 16) by extraction using ether, Et2O, as the solvent. All three compounds are more soluble in ether than water. In your lab, you have aqueous HCl, NaOH, and NaHCO3readily available. (Useful pKas: H2O = 15.7; H2CO3= 6.4)

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 03:50
Which of the following statements about acidic water is true? a. acid has no effect on the h,o molecules. b. the solution contains a larger number of oh ions than h,o ions. c. the solution contains a larger number of h,o ions than qh ions. d. the solution contains an equal number of h,o ions and oh ions. none of the above e.
Answers: 1

Chemistry, 22.06.2019 10:30
Consider the following reactions. (note: (s) = solid, (l) = liquid, and (g) = gas.) mg(s) + ½o2(g) → mgo(s) + 146 kcal/mole h2(g) + ½o2(g) → h2o(g), δh = -57.82 kcal/mole what type of reaction is represented by the previous two examples?
Answers: 3

Chemistry, 22.06.2019 18:10
Given the following at 25c calculate delta hf for hcn (g) at 25c. 2nh3 (g) +3o2 (g) + 2ch4 (g) > 2hcn (g) + 6h2o (g) delta h rxn= -870.8 kj. delta hf=-80.3 kj/mol for nh3 (g), -74.6 kj/mol for ch4, and -241.8 kj/mol for h2o (g)
Answers: 1

Chemistry, 22.06.2019 22:20
Asuspension of yeast cells is being grown under anaerobic conditions such that glucose is degraded to ethanol and carbon dioxide. if one wishes to follow this process by monitoring the release of 14co2, at which positions in the glucose molecule would the 14c label need to be incorporated?
Answers: 2
You know the right answer?
Createa flow chart separating phenol (P, pKa= 10.0), benzoic acid (B, pKa= 4.2), and diphenylmethano...
Questions

Mathematics, 04.12.2020 22:30




Mathematics, 04.12.2020 22:30


Mathematics, 04.12.2020 22:30



English, 04.12.2020 22:30


English, 04.12.2020 22:30

Mathematics, 04.12.2020 22:30

Health, 04.12.2020 22:30

Mathematics, 04.12.2020 22:30


Chemistry, 04.12.2020 22:30



Mathematics, 04.12.2020 22:30