
Chemistry, 02.09.2021 07:00 minecraft37385
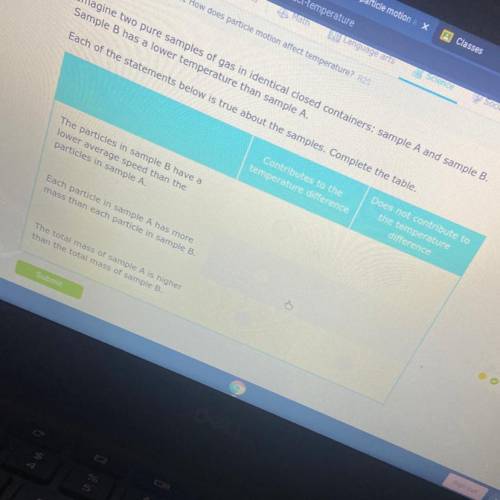
Imagine two pure samples of gas in identical closed containers: sample A and sample B.
Sample B has a lower temperature than sample A.
Each of the statements below is true about the samples. Complete the table.
Contributes to the
temperature difference
Does not contribute to
the temperature
difference
The particles in sample B have a
lower average speed than the
particles in sample A.
Each particle in sample A has more
mass than each particle in sample B.
The total mass of sample A is higher
than the total mass of sample B.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 00:00
The pressure inside a hydrogen-filled container was 2.10 atm at 21 ? c. what would the pressure be if the container was heated to 92 ? c ?
Answers: 2

Chemistry, 22.06.2019 06:40
Which statement correctly describes metallic bonds? a. they form when certain atoms lose electrons and other atoms gain electrons. b. they involve an attraction between anions and cations. they always involvpoth a metal and a nonmetal. d. they can only form between atoms of the same element. e. they form because electrons can move freely between atoms.
Answers: 3


Chemistry, 22.06.2019 19:30
Acetylene gas c2h2 undergoes combustion to produce carbon dioxide and water vapor how many grams of water are produced by the same amount of c2h2?
Answers: 2
You know the right answer?
Imagine two pure samples of gas in identical closed containers: sample A and sample B.
Sample B ha...
Questions

Mathematics, 01.02.2021 17:00




Mathematics, 01.02.2021 17:00

Mathematics, 01.02.2021 17:00

English, 01.02.2021 17:00

Chemistry, 01.02.2021 17:00






Mathematics, 01.02.2021 17:00

Mathematics, 01.02.2021 17:00

Social Studies, 01.02.2021 17:00

Arts, 01.02.2021 17:00





