
Chemistry, 26.08.2021 23:00 FriendlyDude640
Calculate the percentage of limestone that dissolved from each solution. Start by subtracting the final mass from the initial mass. Divide that number by the initial mass. Then multiply the result by 100 to make it a percent. Use this formula:
% dissolved= initial mass- final mass/ initial mass x 100
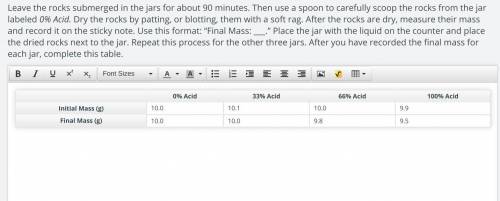
ORIGINAL: 0% Acid 33% Acid 66% Acid 100% Acid
Initial Mass (g) 10.0 10.1 10.0 9.9
Final Mass (g) 10.0 10.0 9.8 9.5
Record the percentage of limestone dissolved in each acid concentration.
Look at attched...

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 06:00
In an investigation that uses the scientific method, which step immediately follows making a hypothesis? o summarizing the results o asking a question o making observations designing an experiment mark this and retum save and exit next submit
Answers: 2

Chemistry, 22.06.2019 07:00
If there is any 12 to 14 girls that need a boyfriend just follow me and let me know
Answers: 1

Chemistry, 22.06.2019 09:00
This chart lists four kinds of polymers and their sources. what can be known about all four polymers, despite their differences? they come from living things. they share ionic carbon bonds. they are at least 100 monomers long. they are made of repeating subunits.
Answers: 2

Chemistry, 22.06.2019 10:30
What determines the average kinetic energy of the particles in a gas? a. the number of collisions b. the number of particles c. the size of the particles d. the temperature
Answers: 1
You know the right answer?
Calculate the percentage of limestone that dissolved from each solution. Start by subtracting the fi...
Questions

English, 25.09.2019 16:30


English, 25.09.2019 16:30

English, 25.09.2019 16:30



Mathematics, 25.09.2019 16:30

English, 25.09.2019 16:30

English, 25.09.2019 16:30

History, 25.09.2019 16:30

English, 25.09.2019 16:30


English, 25.09.2019 16:30



History, 25.09.2019 16:30







