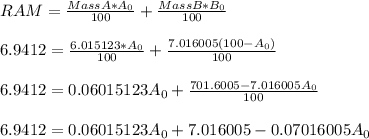Chemistry, 13.08.2021 21:30 mrashrafkotkaat
An certain element Y has two naturally occurring isotopes, 6Y (isotopic mass = 6.015123 amu) and 7Y (isotopic mass = 7.016005 amu). If the atomic mass of the element is 6.9412 amu. What is the percent abundance of 6Y?
Do not include the symbol "%" in your answer.
Report the correct answer to three significant figures

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 18:00
Which three statements represent the benefits of performing experiments using computer simulations?
Answers: 3

Chemistry, 22.06.2019 18:20
Which reason best explains why metals are malleable? a)because they have delocalized electrons b)because they have localized electrons c)because they have ionic bonds d)because they have rigid bonds
Answers: 2

Chemistry, 22.06.2019 22:00
Ill give u brainliest pls how is mass of carbon conserved during cellular respiration
Answers: 1

Chemistry, 22.06.2019 22:30
What is the work done by the electric force to move a 1 c charge from a to b?
Answers: 2
You know the right answer?
An certain element Y has two naturally occurring isotopes, 6Y (isotopic mass = 6.015123 amu) and 7Y...
Questions
















Mathematics, 09.10.2019 04:10


History, 09.10.2019 04:10

History, 09.10.2019 04:10

Mathematics, 09.10.2019 04:10